Publisher
India Pharma Outlook
published at
December 17, 2025
Pharmaceutical Manufacturing in Syria: From Solids to Sterile Injectables with Global Standards
Pharmaceutical manufacturing in Syria is rebuilding around a clear goal: a reliable local supply that meets regional and global quality standards. Syrian companies and their EPC partners are moving from basic formulations to a broader mix of oral solids, liquid orals, sterile injectables, and topical products, guided by WHO GMP pharmaceutical manufacturing frameworks and donor requirements.
For investors, plant heads, and engineering teams, the question is no longer whether capacity exists. The real question is how quickly it can be scaled, made GMP compliant, and prepared for inspections and export.
Overview of Pharma Manufacturing Facilities in Syria and Their GMP Readiness
Pharma manufacturing facilities in Syria range from legacy state-owned plants to newer private factories and retrofit projects that push older assets closer to GMP-ready status. Before the conflict, local manufacturers reportedly produced thousands of types of medicine and met most of the national demand. Today, sanctions, export restrictions, and input shortages limit formal exports, so desktop capacity figures are insufficient.
For any GMP-compliant pharma manufacturing Syria project, sponsors and EPC partners need to look beyond installed equipment and ask:
- What is the real, sustainable run rate of each line?
- Are utilities, quality systems, and documentation aligned with WHO and regional expectations?
- How mature is the workforce in terms of GMP culture and engineering capability?
The answers to these questions determine whether a site can credibly support local hospital tenders, regional supply, or a more ambitious export roadmap.
Oral Solid Dosage Manufacturing Capability in Syria
Oral solids remain the workhorse of pharmaceutical manufacturing in Syria. Many plants are still built around tablets and capsules for branded generics and tender products.
For sponsors evaluating an oral solid dosage manufacturing plant in Syria, the core assessment is simple: can the facility consistently produce tablets, capsules, and coated products at a competitive scale?
Tablets, Capsules, and Coated Products at Scale
A capable oral solid dosage manufacturing plant in Syria will typically include:
- Granulation (wet and/or dry), drying, milling, and blending systems sized for planned batch volumes.
- Compression machines that can hold weight, hardness, and friability within limits during long runs.
- Coating systems that maintain stable spray rates, airflow, and exhaust conditions to protect moisture- or heat-sensitive products.
- Blister and bottle packing lines with in-line checks and sampling plans robust enough for tenders and export buyers.
For a plant head or QA lead, a few simple checks say a lot:
- Do granulation and blending systems scale smoothly from development to commercial batch sizes?
- Are there clear cleaning and line clearance practices to manage cross-contamination risk?
- Can the site show recent process validation and cleaning validation data for key products?
If the answer is yes, the site is on a good path to being a reliable regional supplier of oral solids.
Automation, Containment, and Dust Control
To align solid dose plants with WHO GMP pharmaceutical manufacturing expectations, automation and containment must go beyond simple dust extractors.
Good practice in Syria should include:
- Reduced open handling during dispensing, charging, and discharge through closed transfer systems and well-designed charging ports.
- Zoning and pressure cascades between dispensing, granulation, compression, coating, and packing to control dust and cross-contamination.
- Higher levels of automation in weighing, batch dispensing, data capture, and cleaning verification to support data integrity objectives.
These choices not only help during inspections. They also reduce operator risk, minimize material losses, and make the plant easier to run day-to-day.
Liquid Dosage Manufacturing Facilities in Syria
Liquid oral lines are a visible strength in the Syrian market. Several companies already operate dedicated syrups and suspension lines alongside solid dose and injectable products.
For decision-makers, this means that finding a liquid dosage manufacturing facility that can support mainstream primary care portfolios is realistic, provided that process hygiene and utilities are well controlled.
Syrups, Suspensions, and Oral Solutions
Typical portfolios in Syrian liquid plants include:
- Antipyretics and pain relief products
- Cough, cold, and respiratory syrups
- Antibiotic suspensions
- Paediatric formulations and vitamin tonics
When reviewing a liquid dosage manufacturing facility, focus on:
- How rooms are zoned for dispensing, compounding, filtration, and filling.
- Whether product-contact surfaces (tanks, agitators, transfer lines) use cleanable, compatible materials such as appropriate stainless steels.
- How mixing parameters, piping design, and filtration methods are documented and controlled.
- Microbiological control strategies, including cleaning, sanitisation, hold time limits, and storage conditions.
These factors directly influence batch-to-batch consistency and microbial quality, which are critical if the plant wants to serve tenders or regional export markets.
Mixing, Holding, and Transfer System Design
Mixing, holding, and transfer systems largely define whether a Syrian liquid plant can pass audits or is limited to low-risk markets.
Key design points include:
- Mixing vessels with appropriate agitators, load cells, and temperature control to demonstrate uniformity during validation.
- Holding tanks are sized to minimize long, uncontrolled wait times before filling, reducing microbial risk.
- CIP/SIP coverage to all wetted surfaces, with validated cleaning procedures.
- Transfer lines that are short, drainable, and free from dead legs, with jackets where needed to maintain temperature and discourage microbial growth.
For project sponsors, a liquid plant that demonstrates this level of design discipline is a strong asset when building a multi-dosage site in Syria.
Sterile Injectable Manufacturing: Capability and Constraints
Sterile injectables are central to hospital and emergency care in Syria. Even a limited number of compliant sterile plants can transform access to antibiotics, oncology products, and critical care medicines.
Oubari Pharma was the first private pharmaceutical company in Syria to manufacture injectable medicines in sterilized areas, signalling early investment in sterile capacity.
Asia Pharmaceutical Industries also highlights sterile injectables and biotechnology products as part of its portfolio, indicating that local manufacturers are already working with higher complexity dosage forms.
Several local companies have already invested in sterile capacity, and regional or Indian partners also supply vials, ampoules, and pre-filled syringes into the market. Any sterile injectable manufacturing facility in Syria will therefore be measured against these benchmarks.
Aseptic vs Terminally Sterilized Injectables
Designing or upgrading a sterile injectable manufacturing facility is significantly more complex than adding solids or non-sterile liquids because every step must control viable and non-viable contamination to strict limits.
Sponsors must decide early:
- Which molecules will follow full aseptic processing, using Grade A (ISO 5) filling environments with Grade B backgrounds, typically via isolators or RABS.
- Which molecules can be terminally sterilized in vials, ampoules, or bags, with the focus on sterilizer cycle design and bioburden control?
This decision drives:
- Facility layout and barrier technology
- Environmental classification and monitoring strategy
- Validation scope, including media fills and ongoing cleanroom requalification
- Staffing profiles, especially in process, engineering, and microbiology expertise
Getting this right up front avoids costly retrofits and helps the facility meet WHO GMP, EU GMP Annex 1, and buyer audits.
Cleanrooms, HVAC, and Sterility Assurance
Cleanroom and HVAC design are the backbone of any injectable project aiming to be a credible example of GMP-compliant pharma manufacturing that Syria can offer.
Key design elements include:
- Cleanroom zoning into Grades A, B, C, and D, with clear pressure cascades.
- HEPA filtration, defined air change rates, and localized unidirectional airflow at critical points.
- Qualified HVAC systems with documented performance for pressure differentials, recovery times, temperature, and humidity.
- Integrated design coordination between architects, process engineers, HVAC designers, and CQV teams.
For pharma owners and EPC partners, this is where good pharmaceutical plant design decisions directly reduce regulatory risk and operating costs.
Topical Dosage Manufacturing in Syria
Topical dosage manufacturing in Syria sits at the intersection of pharmaceuticals and a growing cosmetics segment. Local companies already produce a broad mix of medicinal creams, gels, and dermatological products, as well as cosmetic balms and lotions.
For sponsors, this existing know-how can be channelled into a topical dosage manufacturing plant that meets modern GMP expectations.
Creams, Ointments, Gels, and Lotions
A well-designed topical facility in Syria will typically include:
- High-shear mixers or homogenizers, jacketed vessels, and vacuum-capable kettles to create stable emulsions and dispersions.
- Controlled heating and cooling to manage oil and water phase processing and avoid crystallisation.
- Filling lines that can handle a wide range of viscosities without dead zones, using positive-displacement or piston systems.
- Attention to contact materials, ventilation, in-process checks, and container-closure integrity to protect shelf life.
With these features, a topical plant can support both local demand and export-facing portfolios, especially in dermatology and pain relief categories.
GMP Compliance Across Dosage Forms
GMP in Syria is not only about clean rooms. It is about a single, strong quality system that covers every dosage form on site, from tablets and syrups to injectables and creams.
In simple terms, the WHO defines GMP as ensuring products are consistently produced and controlled to standards appropriate for their intended use. For project teams, that translates into a few non-negotiables:
- Standardized procedures for production and quality control across all lines.
- Trained people and qualified equipment, with clear responsibilities and documented controls for cross-contamination and mislabelling.
- Shared utilities designed for the “worst case”, especially pharmaceutical water and HVAC, are sized for the most demanding dosage forms on site.
- Built-in data integrity, so manufacturing and QC records are complete, accurate, and traceable across the product life cycle.
- A capable QC laboratory, in-house or via a qualified partner, following good practices for pharmaceutical quality control labs.
When these foundations are in place, pharma manufacturing facilities in Syria can credibly support local registrations and build towards exports backed by WHO GMP and key pharmacopoeias.
Comparing Dosage Forms by Facility Complexity
The table below helps sponsors compare facility complexity across dosage forms when ranking projects or planning a turnkey pharma plant Syria program.
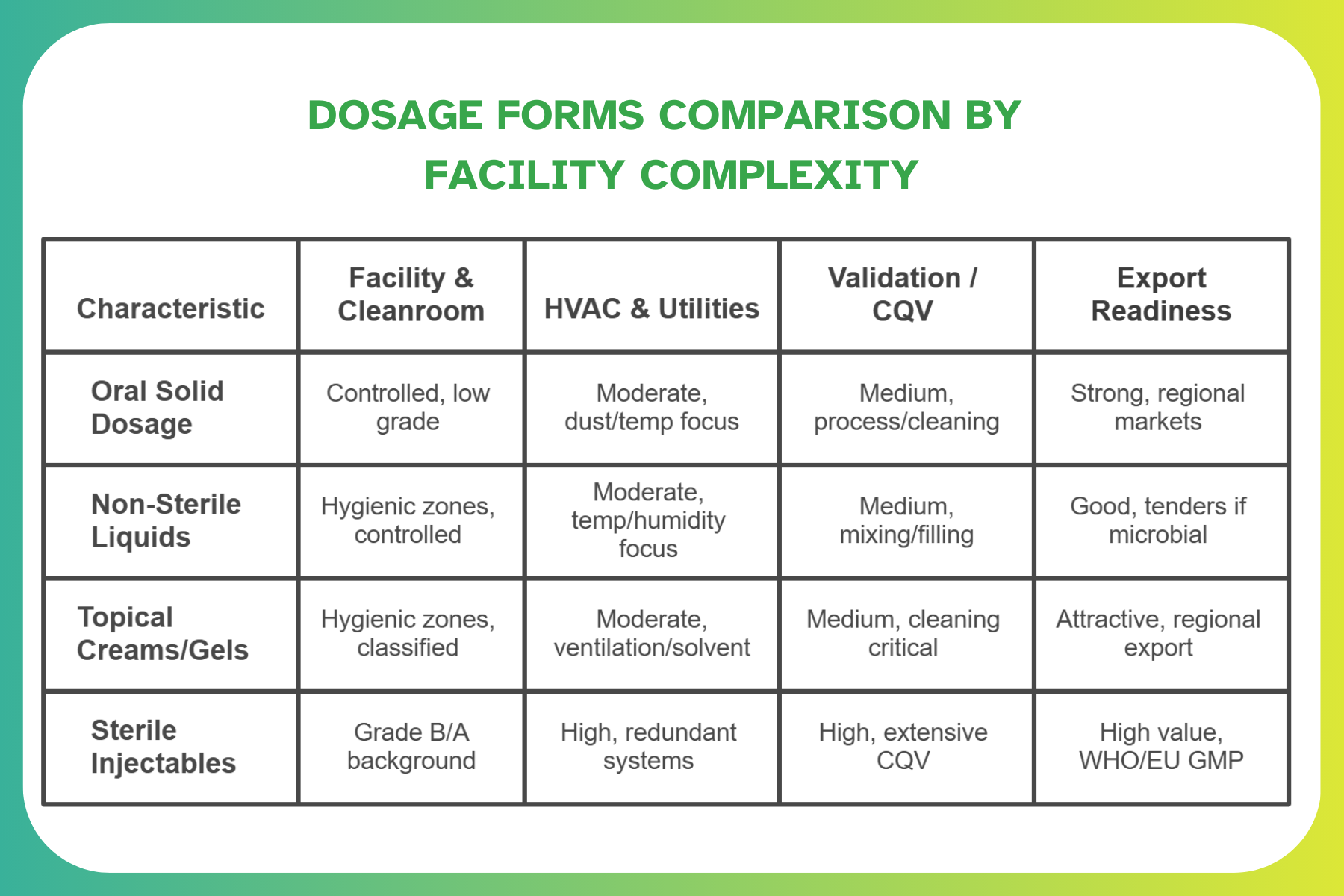
For owners and EPC teams, this comparison clarifies where to start, which lines to stage, and what each step implies for cost, risk, and timeline.
Designing Turnkey, Resilient Pharma Plants in Syria
Syria’s context makes one thing very clear: any new pharma site must be resilient, efficient, and GMP-aligned from the concept stage. For owners, project managers, and EPC partners, this is not about “fighting constraints”. It is about using them as a design brief for better pharmaceutical plant design in Syria projects.
1. Start with an Infrastructure Reality Check
- Treat power stability, water quality, and local maintenance capability as hard design inputs.
- Size and plan utilities (power, pharmaceutical water, HVAC, compressed air, nitrogen, and waste) for the highest-risk dosage forms you want on site.
- Decide early which utilities need redundancy, monitoring, and backup to protect conditions during outages.
2. Link Dosage Forms, GMP Grades, and Utilities
- Match planned dosage forms to what the site can reliably support in utilities, staffing, and QC.
- Define cleanroom grades and zoning for each area, then design HVAC, water, and people/material flows around those needs.
- Use this mapping as the backbone for every turnkey pharma plant project in Syria, so regulatory expectations are built into the layout from day one.
3. Use Retrofits as a Smart Fast-Track
- Retrofitting an existing industrial shell can be faster than a greenfield project when done properly.
- Rework flows of people, materials, and waste to match modern GMP and cross-contamination controls, instead of “fitting equipment where it fits”.
- Prioritize clean, logical routes and dedicated circulation for high-risk and low-risk streams.
4. Plan Turnkey Scope and Sequencing from Day One
- Fix scope, product mix, target markets, and inspection objectives before civil work starts.
- Map shared vs dedicated areas, utility segregation, and cleanroom grades early so architecture, engineering, and procurement stay aligned.
- Write clear URS documents for critical systems and agree on who owns FAT, SAT, IQ, OQ, and PQ across vendors and on-site teams.
5. Build CQV and Constructability into the Design
- Use 3D models to coordinate ducts, pipes, cable trays, and cleanroom envelopes before construction.
- Run constructability reviews so phasing, shutdowns, and tie-ins are planned, not reactive.
- Integrate commissioning, qualification, and validation into the master schedule so the path to the first validation batch is visible for both technical and financial stakeholders.
Handled this way, Syria becomes more than a challenging environment. It presents a clear opportunity to deliver GMP-compliant pharma manufacturing facilities in Syria that are robust by design, inspection-ready, and easier to operate over the long term.
Inotek: Your Strategic Partner in GMP-Ready Multi-Dosage Pharma Facilities in Syria
The complexities of setting up GMP-ready multi-dosage facilities in Syria call for a partner who understands both pharma processes and disciplined project execution. This is where Inotek steps in as your strategic partner. Inotek focuses on designing and delivering pharma facilities aligned with WHO GMP, EU GMP, FDA, and CDSCO requirements, with a particular strength in cleanroom-heavy, regulated environments.
Our comprehensive approach includes:
- Process-led facility and layout design
Translating your product mix across oral solids, liquids, sterile injectables, and topicals into a practical facility layout with clear zoning, people and material flows, and defined cleanroom grades that support inspections and routine operations. - Turnkey project delivery for multi-dosage plants
Managing design coordination, vendor integration, and site execution for turnkey pharma plant projects in Syria, ensuring civil works, utilities, cleanrooms, and process equipment come together as one GMP-ready facility rather than disconnected packages. - Brownfield retrofits and live-site expansions.
Upgrading existing plants with new dosage lines, cleanrooms, and utilities while keeping critical production running, through phased shutdown planning and construction sequencing that reduces disruption and risk. - CQV and documentation aligned to regulators
Integrating commissioning, qualification, and validation into the project from the start, with FAT, SAT, IQ, OQ, and PQ planned and documented in a way that supports WHO GMP and local authority reviews
By partnering with Inotek, pharma manufacturers can see:
- Shorter and more predictable timelines from design freeze to first validation batch
- Fewer surprises during inspections and lower rework on layouts or utilities
- Facilities that are easier to operate day to day, with clear flows and maintainable systems
While compliance is the baseline, successful pharma projects in Syria also need facilities that can cope with local utility realities today and support future growth in demand. Successful GMP-ready pharma facilities in Syria must therefore address growing expectations around:
- Environmental sustainability and energy use
- Supply chain resilience and local infrastructure constraints
- Data integrity, digital monitoring, and long-term maintainability
Inotek helps you design with these factors in mind, so the same site can support capacity ramp-up, additional dosage forms, or export ambitions without major redesign. At Inotek, we ensure your facility is not only audit-ready but also engineered for long-term operational excellence across all dosage forms you plan to manufacture.
Recognized among the Top 10 Pharma Turnkey Contractors and Project Consultants in 2022 and 2025, Inotek helps pharma leaders design, build, and upgrade facilities that meet strict GMP and quality expectations.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you are planning a new facility or optimizing an existing setup, Inotek keeps your project compliant, buildable, and ready to scale.
FAQs
What is the current status of pharmaceutical manufacturing in Syria for oral solids?
Pharmaceutical manufacturing in Syria is rebuilding around local supply and regional demand. Most pharma manufacturing facilities in Syria focus on branded generic tablets and capsules, with retrofits improving utilities and QC labs. A few export-focused plants are moving toward GMP-compliant pharma manufacturing in Syria.
Why are oral solid dosage manufacturing plants important for pharma manufacturing facilities in Syria?
Oral solid dosage manufacturing plants are the backbone of pharma manufacturing facilities in Syria. They produce tablets and capsules for hospital tenders and retail markets. Strong granulation, blending, compression, and coating capability allows Syrian manufacturers to stabilize local supply and build export-ready product portfolios.
What makes a solid dose facility a GMP-compliant pharma manufacturing site in Syria?
A GMP-compliant pharma manufacturing Syria solid dose plant runs under a quality system covering materials, equipment, people, and documentation. Validated processes, qualified utilities, dust control, and data integrity help manufacturers prove consistent tablet and capsule quality during inspections and support tenders or export registrations.
How do turnkey pharma plant projects in Syria support WHO GMP pharmaceutical manufacturing for oral solids?
Turnkey pharma plant Syria projects reduce risk by aligning pharmaceutical plant design, Syria, construction, utilities, and equipment into one integrated scope. For oral solid dosage manufacturing plants, this means faster commissioning, smoother WHO GMP pharmaceutical manufacturing inspections, and commercial supply from an accountable engineering partner.



