Publisher
Inotek
published at
November 19, 2025
Designing Modular Cleanrooms for Scalable CDMO Projects
Struggling to expand cleanroom capacity without disrupting ongoing GMP operations?
You're not alone. CDMOs today face relentless pressure, compressed timelines, multi-product pipelines, rapid tech transfers, and increasingly complex regulatory demands. Yet traditional cleanroom construction often slows you down with extended shutdowns, long build cycles, and validation delays that risk both client onboarding and revenue.
That’s why more CDMOs are shifting to modular cleanroom design, construction, and installation, a faster, more flexible, and GMP-aligned approach to facility expansion.
In this article, we break down how modular cleanrooms help you scale without compromise. From Annex 1-compliant HVAC and ISO 14644 zoning to CQV-ready PODs and brownfield integration, you’ll get a detailed view of the engineering, validation, and execution strategies needed to build smart, scalable CDMO facilities.
Why Modular Cleanrooms Matter for CDMO Scalability
CDMOs operate in an environment defined by volatility, rapid tech transfers, shifting client portfolios, aggressive onboarding timelines, and strict global GMP expectations. Traditional stick-built cleanrooms struggle to keep pace due to long construction durations, extended shutdowns, and rigid layouts.
Modular cleanrooms solve these constraints by offering parallel manufacturing paths, flexible zoning, and predictable GMP commissioning, enabling CDMOs to add capacity or reorient layouts without jeopardising existing operations. For a multi-product CDMO, this translates directly into faster client onboarding, increased batch throughput, and improved asset utilisation.
Fast Deployment With Minimal Production Disruption
Modular cleanrooms are designed for speed without compromising GMP integrity. Their core advantage lies in the off-site fabrication and testing process, which moves the most time-consuming activities out of the live facility.
During installation at the CDMO site, the only activities requiring controlled shutdown windows include:
- Final AHU and utility tie-ins
Integration of HVAC, WFI, clean steam, compressed air, and nitrogen systems into existing utility loops. - Pressure balancing and cascade validation
Ensuring differential pressures align with Annex 1 expectations before equipment loads and staffing commence. - Integration with existing classified zones
Seamless pressure cascade continuation from older structures to new modular segments. - Final validation and EM qualification
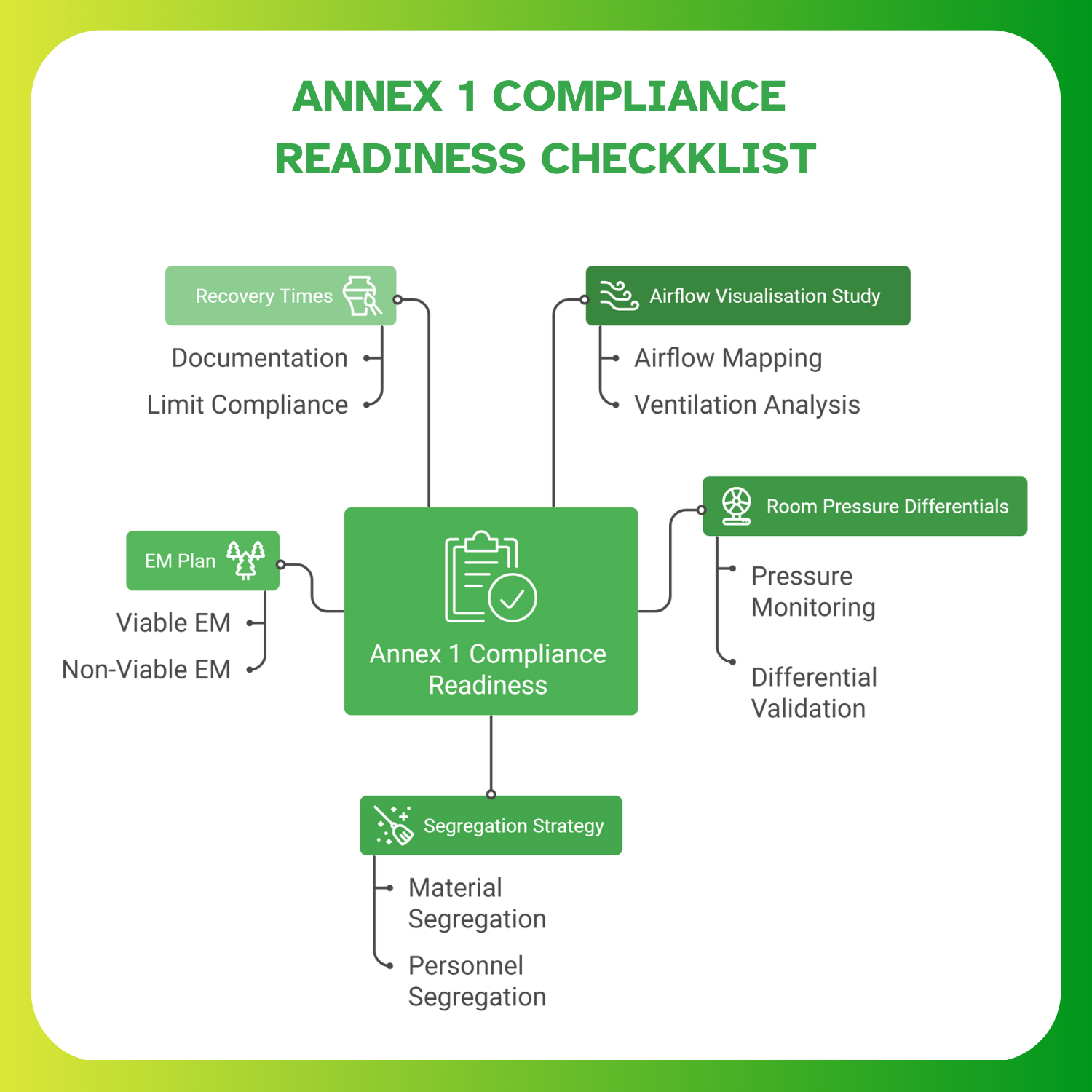
Post-installation steps such as airflow visualisation, non-viable counts, viable sampling, and recovery tests.
Because 70–80% of construction, HVAC setup, and FAT/SAT activities are completed offsite, CDMOs experience dramatically reduced downtime, often contained within a scheduled shutdown window.
This approach protects ongoing GMP batches and reduces the risk of deviations, particularly in facilities operating multiple Grade B or Grade C rooms simultaneously.
Built-in Flexibility for Multi-Product Manufacturing
Product diversity is the operational norm for CDMOs. A single facility may support biologics fill-finish, potent OSD development, and sterile injectables, each with unique zoning, containment, and HVAC requirements. Traditional stick-built spaces are difficult to adapt to such variability.
Modular POD-based systems provide multi-product agility through:
- Fast reconfiguration of zoning
Rooms can be reclassified, merged, or split based on new client processes or updated regulatory expectations. - Plug-and-play expansion
Additional PODs can be added to expand Grade C, Grade D, or support areas without major structural alteration. - Effortless addition or removal of rooms
Changing client pipelines no longer requires disruptive internal demolition or heavy civil work. - Rapid repurposing for new batches or clients
Floor plans can be reshaped to support new process flows, containment levels, or segregated production pathways.
This flexibility helps CDMOs adapt quickly to changing project demands. Modular PODs make it easier to handle faster tech transfers, frequent client turnover, and the need for parallel processing suites without major construction. They also allow cleanroom layouts to be updated rapidly in response to regulatory changes such as Annex 1 revisions, keeping facilities compliant and scalable.
Instead of undertaking year-long renovation cycles, CDMOs can implement reconfiguration projects in weeks, maintaining alignment with client SLAs and commercial timelines.
Key Design Requirements for CDMO Modular Cleanrooms
A CDMO modular cleanroom must do more than install quickly. It needs to integrate seamlessly into existing GMP operations, meet EU GMP Annex 1 and ISO 14644 requirements, support safe flows of personnel and materials, and enable rapid CQV activation. Strong engineering design is what ensures scalability without compromising compliance.
HVAC & Annex 1 Contamination Control
EU GMP Annex 1 places strict expectations on airflow quality, pressure stability, and contamination control.
In a modular environment, the HVAC system must maintain consistent pressure cascades across interconnected rooms and protect room integrity in both at-rest and operational conditions.
Unidirectional airflow in aseptic areas is essential, especially during open product exposure. The system should also recover quickly after door openings or staff movement, preventing particle build-up that could affect product safety.
Equally important is the reliability of AHUs and HEPA filters. Their performance must remain stable under varying equipment loads and occupancy patterns, as fluctuations can directly impact aseptic assurance and environmental monitoring outcomes.
Zoning & Flows for Multi-Product Operations
CDMOs often produce potent OSD, biologics, and sterile injectables in parallel. This diversity creates complex zoning needs that modular cleanrooms must accommodate.
Clear separation of personnel and material movement is essential to avoid unintended crossovers. High-risk and low-risk pathways must remain distinct, and transfer areas should preserve HEPA-filtered integrity during material handling.
Facilities dealing with GMOs, cytotoxic compounds, or allergenic materials require defined containment strategies. Properly designed gowning and degowning areas reinforce directional movement and help maintain classification stability.
Well-organized zoning not only improves contamination control but also creates transparent, audit-ready operational flows that inspectors can easily follow.
Utility Integration for Fast Start-up
One of the strongest advantages of modular cleanrooms is the speed of utility activation.
Pre-engineered utility skids allow rapid integration of WFI, clean steam, compressed air, nitrogen, vacuum, and drainage systems without the long delays associated with traditional builds.
Because these skids are fabricated and FAT-tested offsite, they arrive at the facility with most of the validation already complete. This significantly reduces onsite work, shortens installation by several weeks, and allows the project to move sooner into commissioning and CQV.
For CDMOs with tight onboarding schedules or limited shutdown windows, this rapid activation capability is a major operational advantage.
Timelines, Risk & CQV Considerations
Time-to-ready is one of the biggest drivers for CDMOs. Delays in cleanroom availability directly postpone tech transfers, PPQ batches, and revenue.
Modular vs Traditional Build Timelines
Modular cleanrooms are typically completed in 6–9 months, compared to 14–18 months for traditional builds. This faster delivery accelerates client onboarding, batch scheduling, and commercial readiness.
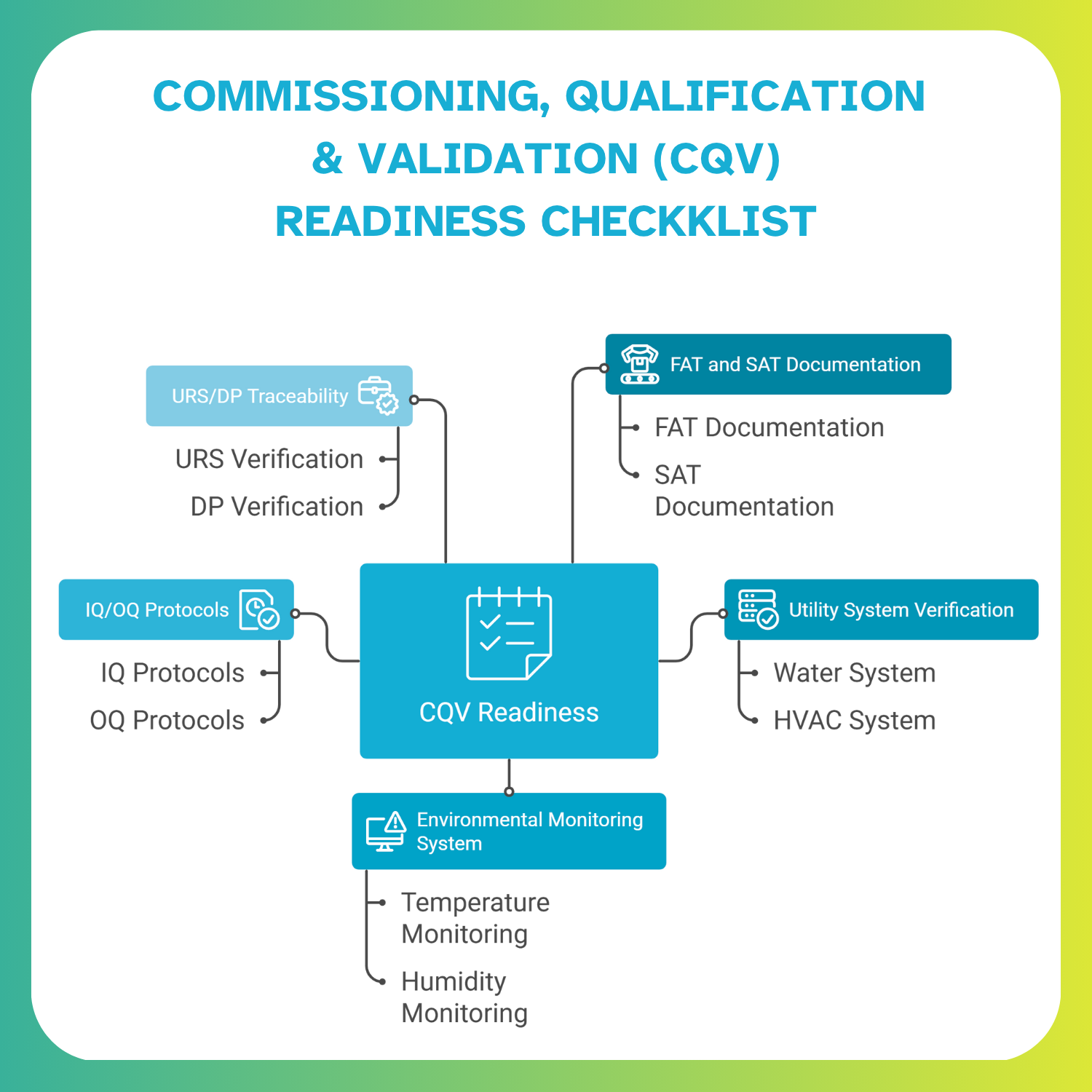
How Modular Accelerates CQV
Because modular PODs undergo FAT and partial SAT offsite, only limited IQ/OQ work is required onsite. This results in shorter commissioning windows, quicker Grade B/A readiness, and fewer start-up deviations.
Brownfield Integration Risks & Mitigation
Integrating modular rooms into live GMP sites can introduce pressure instability, contamination risks, and utility interruptions. These are effectively managed through temporary barriers, phased tie-ins, controlled shutdowns, and portable filtration solutions.
Execution Strategy for CDMO Modular Projects
Successful modular cleanroom delivery depends on strong EPCM alignment. Engineering, QA, validation, and operations must move together, ensuring decisions are locked in early and executed without rework.
Choosing the Right EPCM Partner
The EPCM partner plays a critical role in project success. They must bring Annex 1 expertise, deep cleanroom HVAC design capability, maturity in brownfield execution, and the ability to integrate CQV from URS through OQ and EM. This prevents redesign cycles, avoids compliance gaps, and keeps the project on schedule.
Internal CDMO Decision Milestones
Clean execution depends on disciplined sequencing. URS finalisation, followed by Design Freeze, HVAC Freeze, CQV alignment, and EM strategy closure, ensures that engineering and validation move in sync. This structured approach eliminates late-stage surprises and reduces audit risks.
Tech-Transfer Agility Enabled by Modular Layouts
Modular layouts enhance a CDMO’s ability to onboard new clients quickly. Faster product changeovers, quicker room requalification, and easy segregation of parallel programs shorten downtime and improve throughput. The ability to modify layouts without major construction also supports faster Grade B/A recovery and smoother client transitions.
Engineering Assets Needed for Successful CDMO Modular Projects
Strong engineering documentation is essential for modular cleanroom success. QA teams, inspectors, and validation engineers depend on clear, traceable evidence, so these assets must be developed from the earliest project stages.
Pressure Cascade & Zoning Diagram
A well-defined pressure cascade and zoning diagram helps CDMOs verify airflow directionality, contamination risk control, exposure limits, and Annex 1 compliance. This document becomes a central reference during both design review and validation, ensuring that cleanroom gradients are maintained throughout the facility.
HVAC Integration Map
An HVAC integration map outlines AHUs, prefilters, ducting routes, terminal HEPA locations, pressure control zones, and ISO 14644 classifications. This ensures stable airflow performance during installation, balancing, and CQV. It also helps engineering and validation teams trace system interactions and identify issues early.
Utility Skid Connection Overview
Clear utility connection diagrams help teams integrate WFI, clean steam, compressed air, nitrogen, and drainage points without errors. Because these skids are pre-engineered, accurate documentation accelerates activation and reduces the risk of delays during commissioning.
Inotek: Your Strategic Partner in GMP-Aligned Modular Cleanroom Delivery for CDMOs
The complexities of modular cleanroom design and integration demand specialized engineering, regulatory foresight, and brownfield execution experience, especially when timelines, product mix, and GMP compliance all converge.
This is where Inotek steps in as your strategic partner.
We don’t just install modular units; we engineer fully validated, Annex 1-compliant cleanroom ecosystems that are pre-integrated with HVAC, utilities, and CQV workflows. Every solution we deliver is built for scalability, audit-readiness, and minimal production disruption.
Our Comprehensive Approach Includes:
GMP-First Modular Engineering:
We align every cleanroom build with EU GMP Annex 1, ISO 14644, and client-specific regulatory needs from URS to EM. This ensures faster approvals and zero rework at audit time.
Integrated HVAC Design for Aseptic & Potent Facilities:
Our HVAC systems are purpose-built for biologics, sterile injectables, and potent APIs, maintaining stable pressure cascades and fast recovery across modular zones.
CQV-Embedded EPCM Execution:
We tightly link cleanroom construction with validation timelines, ensuring URS, FAT/SAT, IQ/OQ, and EM readiness are synchronized from Day 1.
Live-Site Expansion & Brownfield Expertise:
We specialize in integrating modular cleanrooms within operational CDMO facilities, limiting shutdowns, managing tie-in risks, and preserving GMP continuity.
Cleanroom Retrofit & POD Reconfiguration Support:
Whether it’s expanding Grade C to B, adding new modules, or adapting to updated Annex 1 zoning expectations, our retrofit team ensures seamless execution without production loss.
By partnering with Inotek, CDMOs have achieved:
- Faster client onboarding and reduced facility ramp-up times
- Fewer regulatory deviations and cleaner EM qualifications
- Flexible layouts that support parallel tech transfers and high client turnover
Building Beyond Compliance
While regulatory alignment is the foundation, future-ready modular cleanrooms must also support evolving CDMO needs around:
- Supply chain agility and high client throughput
- Digital CQV integration and data integrity
- Rapid layout reconfiguration for multi-product operations
At Inotek, we ensure your modular facility isn’t just compliant, it’s built for speed, flexibility, and long-term operational excellence.
Let’s Build What’s Next
🏆 Recognized among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps CDMOs design, build, and retrofit modular cleanrooms that meet the highest GMP and engineering standards.
📞 Connect with our experts today or visitwww.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a new modular suite or expanding within a live site, Inotek ensures your cleanroom project is compliant, resilient, and future-proof.
FAQs
What is a modular cleanroom in pharmaceutical manufacturing?
A modular cleanroom is a pre-engineered, prefabricated cleanroom system designed for fast deployment. It supports GMP compliance, scalable layouts, and quick validation, ideal for CDMOs managing multiple products and tech transfers with minimal operational disruption.
Why are modular cleanrooms preferred by CDMOs?
Modular cleanrooms allow CDMOs to expand capacity faster, with minimal shutdowns. They support multi-product workflows, Annex 1 compliance, and faster CQV readiness, helping CDMOs meet client demands without delaying ongoing manufacturing operations.
How do modular cleanrooms support GMP and Annex 1 compliance?
Modular cleanrooms are designed with stable pressure cascades, controlled airflow, and validated segregation. These features align with EU GMP Annex 1 and ISO 14644, supporting aseptic processing, contamination control, and smooth audit readiness.
What is the typical build time for a modular cleanroom?
Most modular cleanrooms are completed in 6–9 months. This is significantly faster than traditional construction, which can take 14–18 months. The shorter timeline reduces delays in tech transfer, PPQ, and commercial launch.
Can modular cleanrooms be integrated into live GMP facilities?
Yes. Modular cleanrooms are ideal for brownfield sites. With phased utility tie-ins and off-site CQV, they minimize downtime and maintain environmental control, enabling seamless integration into active pharmaceutical manufacturing environments.



