Publisher
India Pharma Outlook
published at
August 27, 2025
What Is the Real Difference Between QA and QC in Pharma GMP?
Have you ever faced an audit query where QA thought QC was responsible, and QC assumed QA had it covered? This blurred accountability is one of the most common—and most costly—compliance risks in pharmaceutical operations. In fact, role confusion between QA and QC has been cited as a contributing factor in over 40% of FDA 483 observations issued to drug manufacturers in 2022
While the difference between QA and QC in pharma GMP is widely recognised in theory, what often goes unresolved in practice is who owns functional responsibilities, risk accountability, and audit sign-offs. This gap creates compliance vulnerabilities that can cascade into regulatory warnings, CAPA delays, and even supply interruptions.
In this article, we decode the QA vs QC in pharma framework within GMP-driven operations, unpack how global regulators define these mandates, and examine why clear ownership models are essential for organisations and project partners delivering compliant facilities.
Why Do QA and QC Boundaries Matter for GMP Compliance?
In GMP-driven pharmaceutical operations, the textbook definitions of Quality Assurance (QA) and Quality Control (QC) are well known:
- QA is preventive, system-level, and governance-focused.
- QC is verification-based, product-specific, and testing-driven.
Yet in the reality of complex manufacturing networks, assigning clear ownership is far more challenging. Boundaries often weaken, giving rise to compliance gaps that expose plants to audit risks.
Consequences of Weak QA/QC Boundaries
When accountability is not defined:
- Audit findings multiply – An EMA inspection trend analysis shows that unclear QA/QC role division is a recurring theme in data integrity and deviation management observations.
- Product quality risks increase – Without clear oversight, out-of-specification results may go untraced to systemic causes.
- Project delays escalate – In turnkey facilities, misaligned roles often force validation packages to be re-worked, delaying go-live dates.
Why Large-Scale Projects Are Especially Vulnerable
In pharma turnkey project management, pharma models, and overlaps become magnified:
- QA may assume QC has validated a process step.
- QC may assume QA owns systemic risk assessment.
- Result: QA/QC overlap issues that undermine the very foundation of GMP compliance.
A PwC life sciences survey found that 56% of delayed facility approvals in emerging markets were linked to unclear QA/QC responsibilities during commissioning and validation.
The Regulatory Reality
Regulators are unambiguous: both QA and QC carry non-negotiable mandates. However, in practice, project teams often struggle with fundamental questions:
- Who signs off on deviations?
- Who owns QA vs QC during GMP audits?
- Who validates corrective actions?
Failure to resolve these leads to FDA 483s, EU GMP observations, and WHO audit remarks—consequences that damage reputation and can block product launches.
Strategic Perspective
For pharma companies and their engineering/project partners, establishing well-defined ownership of QA/QC roles is not an optional governance step—it is a critical enabler of audit readiness and operational continuity. Compliance is not about meeting checklists; it is about designing facilities, processes, and systems that withstand regulator scrutiny from day one.
What Ownership Gaps Between QA and QC Cause Compliance Risks?
In many GMP-driven pharma projects, compliance gaps often arise from unclear handovers between QA and QC. When ownership boundaries are not well-defined, accountability issues surface, leading to audit remarks, CAPA delays, and regulatory non-compliance.
Which QA–QC Handovers Most Often Lead to Compliance Issues?
- Deviations – QA is expected to assess root causes and system-level impact, while QC typically records the initial observation. Without clear ownership, deviations remain unresolved or poorly documented.
- Validation – QC performs laboratory tests during equipment or process validation, but QA ensures protocols align with GMP standards. If blurred, validation packages fail regulatory audits.
- Data Integrity – QC generates and records test data, while QA safeguards systems and reviews processes. If neither owns audit trails or backup controls, data integrity issues surface during inspections.
These overlaps not only weaken GMP compliance but also increase operational costs. Each unresolved ownership gap results in extended CAPA cycles, repeat deviations, and more frequent regulatory findings.
How Do Regulators Like FDA, EMA, and WHO Define QA vs QC Responsibilities?
Global regulators clearly define QA and QC roles in pharmaceutical GMP compliance, leaving little room for ambiguity. Each framework outlines ownership of quality systems, testing, and accountability:
- WHO GMP – QA is responsible for building and maintaining the overall quality system, approving SOPs, and ensuring deviations are addressed. QC confirms product compliance through sampling, testing, and inspection.
- FDA CFR 210/211 – QA holds accountability for procedures, documentation, deviation control, and batch release decisions. QC ensures robust laboratory controls, analytical accuracy, and equipment calibration.
- EU GMP Annex 1 – Adds emphasis on QA’s role in contamination control strategy, while QC owns environmental monitoring and data integrity validation.
While these frameworks are precise, challenges emerge when pharma companies or GMP turnkey project management pharma providers fail to translate regulatory intent into internal ownership structures. Without this alignment, gaps in accountability often lead to audit surprises, regulatory observations, and product quality risks.
To stay compliant, companies must map QA and QC roles in pharmaceutical industry mandates directly into their facility design, validation, and commercial production processes.
While global frameworks clearly define QA and QC, it’s often difficult for teams to see these differences side by side. To make this clearer, here’s a one-page QA vs QC comparison table you can use as a quick reference during audits or team training.
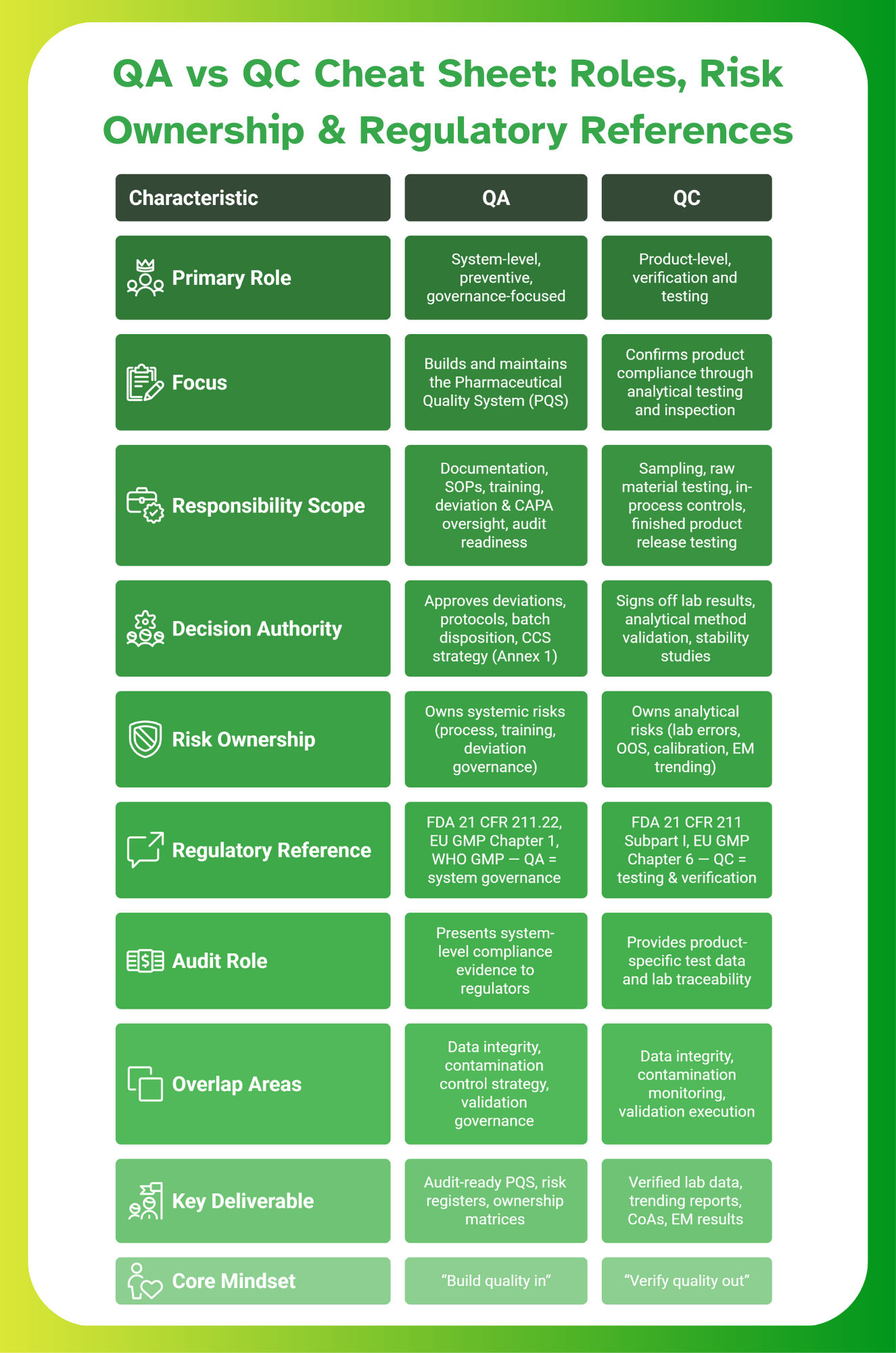
Who Owns What in GMP Projects: QA or QC?
In modern pharma projects, particularly those executed by a pharma turnkey solution provider, clarity on QA/QC risk ownership in pharma plants is critical. QA owns process-level compliance, while QC owns product-level verification. When these roles are not clearly mapped, functional ownership in pharma turnkey projects gets diluted. The result: extended project timelines, engineering rework, and weakened compliance accountability.
A structured ownership model ensures that responsibilities move seamlessly across stages — from design to validation to audit readiness. For companies relying on pharma turnkey engineering solutions, this clarity prevents overlap, avoids audit finger-pointing, and secures faster regulatory approvals.
Who Signs Off Documentation and SOPs—QA or QC?
- QA: Owns SOP development, documentation approval, and GMP alignment.
- QC: Advises on sampling, test requirements, and method suitability but does not influence system design.
👉 In turnkey projects, this distinction ensures QA drives governance, while QC prepares test readiness. Clear sign-offs here prevent downstream disputes about ownership of protocols or test methods.
Who Owns Commissioning and Validation Responsibilities in GMP Projects?
- QA: Ensures protocols, reports, and validation processes meet GMP standards.
- QC: Executes laboratory testing, validates analytical methods, and confirms data accuracy.
👉 A GMP turnkey project management pharma partner must create a Validation Master Plan integrating both functions. Without this clarity, validation packages often face rejection during regulatory review.
Who Is Accountable During GMP Audits: QA or QC?
- QA: Owns overall compliance accountability and system-level sign-offs.
- QC: Owns laboratory records, test accuracy, and analytical traceability.
👉 Regulators often ask, “Who owns QA vs QC in GMP audits?” Companies must show a defined audit ownership matrix. In a pharma facility turnkey project provider model, every audit finding must map to the right owner — QA for systemic gaps and QC for analytical issues.
How Should QA and QC Split Risk Ownership in GMP Operations?
In GMP operations, compliance gaps often arise not from missing procedures but from unclear QA/QC risk ownership in pharma plants. To avoid duplication and unmanaged risks, QA and QC must divide accountability with precision. For a pharma turnkey project company, this becomes even more critical — since handovers between engineering, QA, and QC occur under compressed project timelines.
By defining risk ownership early, companies reduce CAPA delays, avoid regulatory findings, and secure faster plant approvals. A strong framework ensures that every non-conformance, test failure, or system deviation has a clearly accountable owner.
What Risks Belong to QA vs QC?
- QA: Owns systemic risks — SOP failures, inadequate training, deviation handling, and GMP process compliance.
- QC: Owns analytical and testing risks — incorrect results, stability study failures, and equipment calibration errors.
👉 Example: When an out-of-specification (OOS) result occurs, QC investigates test validity while QA ensures systemic controls are adequate. This division prevents duplication and ensures no risk is overlooked.
What Risks Must QA and QC Jointly Own?
Some risks must be jointly owned in pharma turnkey engineering solutions:
- Contamination events during commissioning – QC monitors results, while QA ensures systemic corrective actions are applied.
- Validation errors – QC confirms test accuracy, while QA checks protocol compliance.
👉 If either side fails, the turnkey partner risks audit citations. Shared accountability prevents teams from pushing ownership back and forth.
How Can Risk Registers Help Clarify QA–QC Ownership?
A practical tool for managing accountability is a risk register, which lists:
- Potential risks
- Probability and impact
- Assigned owner (QA or QC)
In turnkey projects, risk registers also capture engineering-related risks — e.g., vendor qualification gaps or equipment installation errors — and link them to QA/QC oversight.
By maintaining a risk register, companies demonstrate transparency to regulators. It proves that QA/QC risk ownership in pharma plants is clearly mapped into CAPA loops, supporting both compliance and continuous improvement.
Which Regulatory Mandates Define QA and QC Boundaries?
Clear ownership of QA and QC is not simply an efficiency matter — it is a regulatory expectation. Global GMP frameworks from WHO, FDA, and EU all define QA/QC roles with explicit mandates. For pharma companies and GMP turnkey project management pharma partners, aligning internal practices with these rules is essential to avoid audit citations and secure timely project approvals.
While each region’s framework reinforces the same principle — QA governs the system, QC confirms the product — their emphasis differs across responsibilities such as contamination control, data integrity, and product release.
WHO Guidance on QA/QC
According to WHO GMP guidelines:
- QA: Builds the quality system, approves SOPs, ensures deviations are addressed, and owns final product release decisions.
- QC: Conducts sampling, testing, and stability studies, ensuring results meet approved specifications.
👉 For turnkey projects, WHO guidance ensures plant handovers are not only technically complete but also fully compliant with the quality system.
FDA vs EU Mandates
- FDA CFR 210/211: QA is accountable for documentation, deviation control, record-keeping, and batch disposition. QC owns laboratory controls, equipment calibration, and analytical accuracy.
- EU GMP: While reinforcing QA’s role in system ownership, it places stronger emphasis on contamination control, particularly in sterile manufacturing facilities.
👉 A pharma turnkey engineering solutions partner must adapt ownership models to regional nuances — there is no one-size-fits-all approach.
What Does EU GMP Annex 1 Say About Data Integrity Ownership?
The 2022 EU GMP Annex 1 update heightened expectations around data integrity:
- QA: Ensures electronic records are validated, audit trails secured, and review systems maintained.
- QC: Guarantees test data is accurate, traceable, and complete.
👉 For turnkey projects, data ownership must be embedded at the commissioning stage, not patched later. A pharma facility turnkey project provider that integrates Annex 1 requirements early avoids rework, audit remarks, and costly non-compliance risks.
How Do Engineering, QA, and QC Intersect in Turnkey Pharma Projects?
In modern GMP projects, engineering teams play a direct role in quality outcomes. It is no longer sufficient for engineers to simply “hand over” a facility. Regulators now expect Good Engineering Practice (GEP) and Construction Quality Management (CQM) to be integrated with QA and QC functions from the very beginning.
For a pharma turnkey project company, this intersection is critical:
- Engineering defines the foundation.
- QA ensures compliance with GMP and system integrity.
- QC verifies product readiness through testing and validation.
When these functions operate in silos, the result is requalification cycles, project delays, and higher costs. When integrated, facilities are delivered faster, cleaner, and fully audit-ready from day one.
What Does GEP Require—QA or QC Ownership?
Engineering teams carry responsibility for risk-based commissioning under GEP. This includes:
- Verifying installation and utility performance against design intent.
- Documenting commissioning results in a way that QA can approve and QC can rely on.
👉 In turnkey execution, ownership is mapped as:
- Engineering: Executes installation and commissioning.
- QA: Confirms compliance with GMP standards.
- QC: Validates analytical results and environmental outcomes.
This shared ownership ensures commissioning is not merely mechanical but aligned with GMP expectations.
How Does CQM Strengthen QA and QC Boundaries in GMP Projects?
CQM ensures that design intent is preserved during construction. Within a pharma turnkey delivery model, it serves as the bridge between site execution and quality compliance.
- Engineering applies CQM to confirm installations match design drawings and specifications.
- QA reviews documentation to ensure CQM practices align with GMP principles.
- QC validates through environmental monitoring and material quality checks.
👉 By embedding CQM, a pharma turnkey engineering solutions partner eliminates gaps between construction quality and regulatory quality — one of the most common root causes of audit findings.
What Ownership Challenges Do Turnkey Pharma Projects Face?
Turnkey pharma projects promise speed, integration, and single-point accountability. But in GMP-driven operations, ownership of QA and QC cannot be diluted into contracts alone. A pharma turnkey project company must balance efficiency with compliance clarity, ensuring that regulators still see clear QA/QC ownership within the client organisation.
Without robust governance, companies risk overlapping responsibilities — or worse, ownership voids that trigger audit findings and inspection failures.
What Are the Benefits of Clear QA–QC Accountability in Turnkey Projects?
When executed with the right governance, turnkey delivery models offer significant advantages:
- Single-contractor accountability → faster resolution of design or execution issues.
- Reduced project delays → integrated teams prevent bottlenecks.
- Stronger alignment → engineering, QA, and QC operate under one unified framework.
👉 For pharma facilities, this means smoother commissioning, quicker qualification, and fewer audit surprises.
Where Do QA–QC Ownership Risks Appear in Turnkey Contracts?
If responsibilities are not clearly defined, turnkey delivery creates compliance ambiguity:
- Engineering assumes QA/QC tasks without formal approval.
- QC findings remain unresolved because the contractor treats them as client-owned.
- Audit ownership gaps emerge when regulators ask: “Who approved this system?”
👉 These risks can undermine the value of turnkey execution. Without contractual clarity, even the best-designed facilities may fail inspection.
What Best Practices Keep QA and QC Aligned in GMP Projects?
To retain turnkey efficiency while ensuring compliance, solution providers for pharma plants and client QA/QC teams should:
- Define ownership explicitly in contracts and project charters.
- Use RACI matrices to map roles across design, commissioning, and validation.
- Establish joint QA/QC review boards for high-risk project stages.
👉 This balanced governance model ensures that turnkey projects deliver both speed and audit readiness, protecting timelines and regulatory approvals.
How Can Turnkey Models Ensure QA–QC Ownership Clarity?
At Inotek, we recognise that in pharma projects, QA vs QC ownership is not optional — it is the foundation of audit readiness. As a trusted pharma turnkey facility project provider, we go beyond design and construction by embedding compliance ownership models into every stage of delivery.
Our approach ensures:
- Clear QA/QC boundaries from day one – QA governs the system, QC validates the product.
- Audit ownership matrices – showing exactly who signs off on deviations, protocols, and CAPA loops
- Integrated engineering, QA, and QC reviews – preventing overlap issues in validation, commissioning, and inspections.
By aligning with WHO GMP, FDA CFR 210/211, and EU GMP Annex 1, Inotek guarantees that facilities are not only technically sound but also regulator-ready. Clients benefit from faster approvals, reduced risk of 483s or audit findings, and smoother commercial launch.
For pharma companies and project consultants, this means one thing: turnkey delivery without compliance compromise. Inotek ensures that every project is built right, documented right, and owned right — with no ambiguity when the auditor arrives.
Inotek: Your Strategic Partner in QA/QC Ownership for GMP Turnkey Projects
The complexities of defining QA vs QC ownership in GMP-driven pharma projects demand specialised expertise and compliance-focused execution. This is where Inotek steps in as your strategic partner. We don’t just deliver facilities; we engineer audit-ready systems, documentation governance, and risk ownership models into every project—aligned with global regulatory standards like FDA, EMA, WHO, and CDSCO.
What Solutions Establish Clear QA–QC Ownership from Day One?
- QA/QC Ownership Mapping – Building clear accountability matrices so that audit responsibilities and risk ownership are never ambiguous.
- Validation & Commissioning Integration – Ensuring QA protocols and QC test verifications flow seamlessly across project stages to avoid regulatory rejections.
- Audit-Ready Documentation Systems – Embedding SOP governance, deviation handling, and CAPA tracking into turnkey delivery.
- Engineering–Quality Synergy – Integrating GEP (Good Engineering Practice) and CQM (Construction Quality Management) with QA/QC to eliminate compliance gaps.
- Digital & Data Integrity Support – Implementing Annex 1–aligned systems for secure audit trails, electronic records, and quality dashboards.
What Outcomes Result from Clear QA–QC Ownership?
By partnering with Inotek, pharma manufacturers have achieved:
- Faster facility commissioning timelines with zero requalification delays.
- Reduced regulatory gaps through audit ownership matrices and risk registers.
- Lower CAPA cycle times by embedding QA/QC accountability from design to operation.
- Smoother global approvals across US FDA, EU GMP, and WHO inspections.
How Do Future GMP Trends Influence QA and QC Boundaries?
While compliance forms the foundation, successful turnkey projects must also address rising industry demands for:
- Environmental sustainability in facility design and operation.
- Supply chain resilience through robust vendor qualification and documentation trails.
- Digital compliance with validated electronic systems and real-time data integrity.
At Inotek, we ensure your pharma facility isn’t just audit-ready, but engineered for long-term operational excellence.
How Can You Build Audit-Ready QA–QC Ownership Today?
Recognised among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma leaders design, build, and upgrade facilities that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a greenfield facility or optimising an existing setup, Inotek ensures your project is compliant, resilient, and future-proof.
FAQs
How do regulators define QA vs QC in pharma GMP?
Regulators like the WHO, FDA, and EU GMP define QA as system-level accountability and QC as product-level verification. QA governs SOPs, documentation, and contamination control, while QC owns testing, data accuracy, and environmental monitoring.
What causes compliance risks in pharma GMP projects?
Most compliance risks stem from unclear QA/QC ownership during deviations, validation, and data integrity. Without defined responsibility, CAPA cycles lengthen, and audits expose repeat regulatory findings.
Who owns QA vs QC responsibilities in pharma GMP projects?
QA owns process-level compliance (SOPs, GMP alignment, audit accountability), while QC owns product-level verification (testing, validation accuracy, data integrity). In turnkey projects, clear ownership prevents delays, overlaps, and audit findings.
Why is QA/QC risk ownership important in pharma GMP projects?
Clear QA/QC risk ownership ensures systemic and analytical risks are assigned to accountable teams. This reduces CAPA delays, prevents duplication, and helps pharma turnkey projects pass regulatory audits without findings.
What are QA and QC responsibilities under GMP regulations?
Under GMP, QA governs quality systems, documentation, deviations, and product release, while QC verifies product compliance through sampling, testing, and data accuracy. Regional frameworks (WHO, FDA, EU GMP) emphasise different ownership areas like contamination control and data integrity.
What is the role of engineering in GMP turnkey pharma projects?
Engineering teams ensure Good Engineering Practice (GEP) and Construction Quality Management (CQM) are integrated with QA and QC. This alignment preserves design intent, reduces audit risks, and ensures facilities are delivered fully compliant from day one.
What are the risks of turnkey delivery in pharma GMP projects?
Turnkey pharma projects risk audit ownership gaps if QA/QC responsibilities are unclear. Without defined governance, engineering may assume QA/QC tasks, QC findings go unresolved, and facilities risk failing inspections.



