Publisher
Inotek
published at
December 10, 2025
How GMP Compliance Can Drive the Rebuilding of Syria’s Pharmaceutical Industry
Can Syria’s pharmaceutical industry recover from the devastation caused by years of conflict? The country’s once-thriving pharma sector, which supplied most of its domestic drug needs and exported medicines across the region, now faces crippling infrastructure challenges. Rebuilding this vital industry is not just about restoring facilities; it’s about ensuring that the drugs produced are safe, high-quality, and globally compliant.
Good Manufacturing Practice (GMP) standards provide the blueprint for this recovery. By adhering to GMP regulations, Syria can establish a sustainable pharmaceutical manufacturing ecosystem that ensures efficiency, safety, and regulatory compliance. This article explores how GMP compliance plays a critical role in Syria's path to rebuilding its pharmaceutical infrastructure, addressing the challenges of post-conflict recovery, and re-establishing trust in the local drug supply.
The Current State of Syria’s Pharmaceutical Industry: Challenges & Opportunities
Before the conflict, Syria had a strong pharmaceutical manufacturing industry, meeting approximately 90% of its domestic drug needs through local production. The country's pharmaceutical sector was one of the largest in the region, exporting medicines to neighboring countries. Representative manufacturers in the ecosystem include Masters Pharma, Ibn Al-Haytham Pharmaceutical Industries Co., and Maatouk Group, reflecting the sector’s historic breadth and capacity.
However, the war devastated infrastructure, leading to significant gaps in local drug production and severe disruptions to the supply chain. While many pharmaceutical plants were destroyed, this challenge also presents an opportunity: Rebuilding Syria’s pharmaceutical industry in accordance with Good Manufacturing Practice (GMP) this could restore local drug production, reduce reliance on imports, and restore supply chain resilience in the long term.
The Impact of Conflict on Pharma Infrastructure in Syria
The Syrian conflict has severely impacted the country's pharmaceutical infrastructure, with widespread destruction of facilities. Key manufacturing zones, particularly in Aleppo, Homs, and Damascus, faced extensive damage. Many pharmaceutical factories were either completely destroyed or rendered non-functional, leaving the nation without a significant portion of its previous drug production capacity. In some regions, up to 90% of local production was halted, disrupting the supply of essential medicines, including treatments for chronic conditions, vaccines, and life-saving injectables.
The collapse of these production facilities, combined with disruptions to storage and transportation networks, led to widespread shortages of medicine, especially in the northern and southern provinces. Local access to essential drugs became limited, leading to dependence on smuggled or unregulated medicines, which often posed significant health risks due to their unknown origins or safety profiles.
This destruction highlights the critical need for GMP-compliant infrastructure to ensure drug quality, safety, and sustainability for future production in Syria.
Regulatory Gaps in Syria’s Pharma Sector
Before the war, Syria’s pharmaceutical regulatory system faced challenges due to limited oversight and outdated compliance mechanisms. The conflict only exacerbated these issues. With the collapse of key institutions and the loss of qualified personnel, regulatory oversight in Syria became almost non-existent in certain areas, further hindering efforts to maintain high-quality drug production standards. For manufacturers like Star Health Group and THERIAC Pharma, strengthened, transparent GMP oversight is central to rebuilding market confidence and safe supply.
The country’s regulatory frameworks were already struggling to enforce standards on raw material imports, manufacturing processes, and distribution systems. During the war, sanctions, economic challenges, and disrupted supply chains made it difficult to guarantee the quality of medicines produced or imported.
The absence of a strong regulatory system led to an influx of unregulated drugs into the market, raising concerns about their safety, efficacy, and authenticity.
Why GMP Compliance Is Crucial for Rebuilding Pharma Manufacturing in Syria
Good Manufacturing Practice (GMP) is the internationally recognized system ensuring that medicines are consistently produced and controlled to quality standards appropriate for their intended use. For companies such as Well Health Pharma, VITA Pharmaceutical Industry, and Miamed Pharmaceutical Industries, aligning with internationally recognized GMP frameworks is essential to ensure batch consistency, safety, and export readiness.
Adopting GMP compliance is essential for Syria as it rebuilds its pharmaceutical infrastructure, ensuring that locally produced drugs meet international quality standards and safety and efficacy expectations. GMP ensures that production systems are designed to prevent contamination, enforce traceability, and maintain consistency in every batch, all crucial requirements when restarting manufacturing in a post-conflict environment.
Key Elements of GMP Compliance for Pharma Facilities
GMP compliance is built upon several core elements that manufacturers must implement to ensure medicines are safe and consistent.
- Quality Management System (QMS): The backbone of GMP, governing all aspects from documentation and change control to corrective/preventive actions.
- Personnel: All staff must be properly trained, qualified, and have clearly defined roles and responsibilities.
- Facilities & Equipment: Manufacturing areas, storage, utilities (e.g., water systems, HVAC), and cleanrooms (if required) must be designed, qualified, and maintained to prevent contamination.
- Sanitation & Hygiene: Both personal and environmental hygiene are mandatory. Regular cleaning, waste disposal, and verified sanitation protocols are essential to avoid cross-contamination.
- Documentation & Record-Keeping: Batch records, SOPs, lab test logs, deviation reports, and cleaning logs must be accurate and maintained for traceability throughout the production process.
- Packaging & Labeling: Packaging materials and processes must be controlled and validated. Labels must accurately reflect the contents, strength, expiry, and storage instructions.
Together, these elements form the backbone of GMP compliance, ensuring that quality is embedded into every step of the process, not checked only at the end.
Manufacturers, including Ultra Medica Pharmaceutical Industries and Oubari Pharma, benefit most when QMS, documentation, training, and sanitation are embedded as daily practice, rather than treated as periodic projects.
Infrastructure Needs for GMP-Certified Pharma Plants
Meeting GMP standards requires more than just equipment; it also involves facility design, environmental control, and validated processes.
- Facility Layout: Manufacturing premises must be laid out to prevent contamination and cross-contamination. This often involves segregated production zones, controlled cleanrooms (for sterile or sensitive dosage forms), and validated cleaning protocols.
- Equipment: Equipment must be correctly installed, qualified (IQ/OQ/PQ), maintained, and cleaned per documented procedures.
- Quality-Oriented Workflow: The facility should support material flow, personnel flow, waste disposal, cleaning, and sampling, all designed to minimize mix-ups or contamination.
For post-conflict rebuilding in Syria, this means that reconstruction or retrofit efforts must incorporate GMP-grade facility design from the very beginning. Otherwise, even structurally sound buildings may fail to deliver safe, compliant pharmaceutical products.
Risks of Non-Compliance with GMP in Pharmaceutical Manufacturing
The risks of non-compliance with GMP are significant and well-documented:
- Product Recalls & Legal Consequences: Non-compliance can lead to recalls, legal consequences, and serious threats to patient safety.
- Financial & Reputational Damage: A high-profile GMP failure can cost over $1 billion, highlighting the severe financial and reputational risks of ignoring GMP standards.
- Health Risks: Inadequate environmental monitoring, poor documentation, insufficient cleaning or validation, and unqualified personnel have repeatedly led to contamination, sterility failures, and product defects.
- Loss of Public Trust: Beyond direct recalls and shutdowns, non-compliance undermines public trust in the local drug supply. In a post-conflict context like Syria, where rebuilding credibility is critical, failure to adhere to GMP can lead to reliance on unregulated, potentially unsafe medicines, putting patients at risk and hindering efforts to restore a stable, trustworthy pharmaceutical industry.
For Syria’s pharmaceutical rebuilding efforts, merely reconstructing buildings is insufficient. For any manufacturer, whether Star Health Group or Sedanah Trading Company, non-compliance raises recall, reputational, and supply-continuity risks that are particularly costly in a rebuilding context.
GMP-compliant infrastructure, including equipment, facility design, environmental controls, and validated systems, is critical to producing safe, high-quality medicines that meet international standards. Only then will Syria be able to regain public trust, ensure patient safety, and re-enter global pharmaceutical supply chains.
Building GMP-Compliant Facilities for Pharma Production in Syria
Rebuilding Syria’s pharmaceutical infrastructure requires a strategic approach to facility design and construction. Adhering to Good Manufacturing Practice (GMP) standards ensures that production systems are safe, efficient, and capable of delivering high-quality medicines.
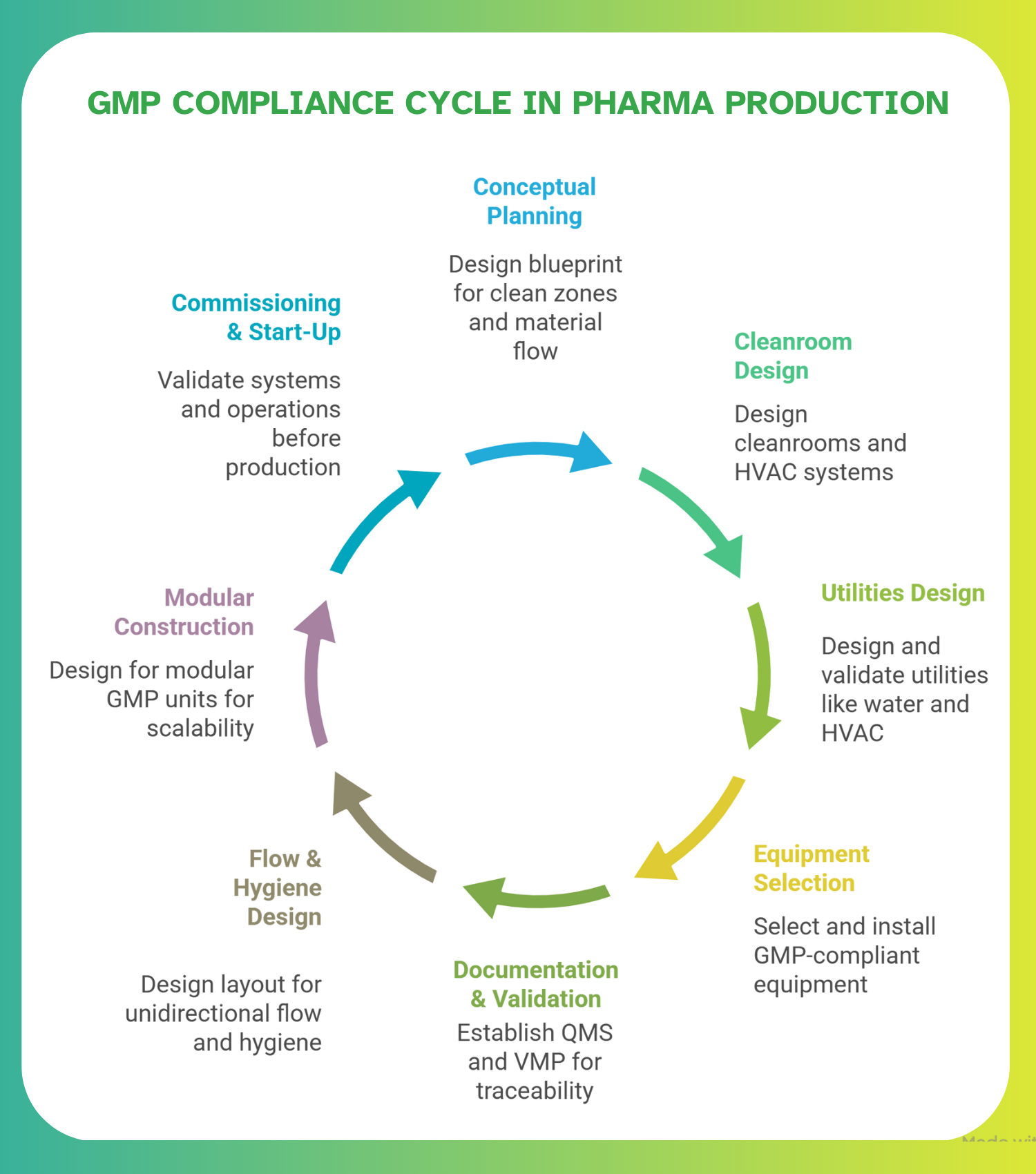
Conceptual & Layout Planning: Flow, Zoning & Barrier Design
Early-phase design blueprint layout of zones, segregation of clean vs. non-clean areas, material flow vs. personnel flow, barrier systems, and planning for future scaling. Designing the layout correctly up front reduces the risk of cross-contamination and ensures compliance from day one. Guidance documents recommend comprehensive planning early to freeze layout, material/personnel flow, utilities and utility routes, room data sheets, and technical specs before detailed engineering begins.
Cleanroom & Controlled Environment Design (Zoning, HVAC, Airflow, Cleanroom Grades)
Cleanroom classification (grades A–D or ISO equivalents), HVAC design and air handling, pressure differentials, air-change rates (ACH), airflow patterns (unidirectional vs. turbulent), environmental controls (temperature, humidity, filtration), critical to contamination control and required for sterile or sensitive dosage forms. Standards and guidance from organizations like the World Health Organization (WHO) and broader GMP-HVAC guidelines inform this design.
Utilities & Infrastructure: Water, HVAC, Utilities Validation, Support Systems
Design and qualification of utilities needed for pharma manufacturing, purified water systems, compressed air, HVAC, clean utilities, waste handling, drainage, sanitation, and utilities routing. GMP guidance requires utilities to meet defined standards for safety, cleanliness, and maintenance.
Equipment Selection, Installation & Qualification (IQ/OQ/PQ), Cleanability & Maintenance
Selecting manufacturing and support equipment that meets GMP requirements (e.g., smooth surfaces, non-reactive, easy to clean/sterilize), correct installation, and full qualification through Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Ensures reproducibility, compliance, and audit readiness.
Documentation, Validation & Quality Systems Integration (QMS, VMP, Monitoring)
Establishing a Quality Management System (QMS), drafting a Validation Master Plan (VMP), procedures for environmental/microbial monitoring, cleaning/disinfection protocols, SOPs, change control, batch documentation, all necessary to ensure ongoing GMP compliance, traceability, and audit readiness. Validation and documentation are central to GMP compliance.
Material and Personnel Flow, Hygiene & Contamination Control Procedures
Layout and process design to ensure unidirectional workflow (materials from raw-material reception → manufacturing → QC → packaging → dispatch), separation of clean and dirty zones, gowning rooms/airlocks, personnel hygiene, cleaning procedures, waste material handling to avoid cross-contamination or mix-ups.
Modular or Phased Construction & Scalability Considerations
Designing the facility with modular GMP-ready units or phased expansion in mind, allowing incremental rebuild or expansion, flexibility for varied dosage forms, scalable utilities, and cleanroom suites. This is especially relevant in a reconstruction context where resources, budgets, or demand may evolve.
Commissioning, Qualification & Start‑up (Commissioning Plan, Validation, Audit/Inspection Readiness)
Final phase before production, validating that facility, systems, utilities, and operations meet GMP standards; executing commissioning, IQ/OQ/PQ, environmental monitoring, qualification of cleanrooms & utilities; preparing documentation for regulatory or client audits; ensuring full compliance before manufacturing begins.
The Strategic Benefits of GMP Compliance in Rebuilding Syria’s Pharmaceutical Industry
Implementing Good Manufacturing Practice (GMP) standards during the rebuilding of Syria’s pharmaceutical infrastructure offers more than just a compliance checklist; it delivers strategic value in quality, safety, efficiency, credibility, and market access.
Operational Efficiency and Cost Benefits of GMP Compliance
GMP compliance establishes robust systems for quality control, traceability, and process control, reducing risks of contamination, mix-ups, and rejects.
By minimizing batch failures, waste, and rework, GMP helps avoid costly recalls and reduces losses associated with defective products.
Standardized procedures and quality systems lead to predictable, reproducible manufacturing, streamlining operations, reducing downtime, and improving throughput.
Over the long term, these efficiencies often generate a favorable return on investment (ROI), as cost savings from reduced waste, rework, recalls, and regulatory penalties accumulate.
In a rebuilding context like Syria, such operational efficiency and cost control translate into more reliable local production of medicine, helping make treatments affordable and sustainable for the population.
Market Credibility and Competitiveness with GMP Certification
GMP-certified facilities signal compliance with internationally accepted quality and safety standards. This helps build trust among regulators, healthcare providers, and patients.
For export-oriented ambitions, GMP compliance is often a mandatory requirement in many importing countries. Without it, products may be rejected or barred from entry.
By meeting GMP standards, Syrian pharmaceutical companies can access regulated global markets, attract international partnerships, and contribute to economic recovery.
GMP compliance also differentiates a plant in the competitive pharma landscape. It becomes a selling point for partnerships, contract manufacturing, and investor confidence, especially relevant for firms looking to scale or collaborate internationally.
Overall, GMP certification can reposition Syria's pharmaceutical capacity from a domestic, fragmented rebuild to a credible, export-ready pharmaceutical manufacturing base capable of competing internationally.
Ensuring Long-Term Trust in the Local Drug Supply
Ensuring Long-Term Trust in the Local Drug Supply. GMP compliance helps rebuild public and institutional trust in the local drug supply. In a post-conflict context like Syria’s, where supply disruptions and quality concerns may persist, having a transparent, GMP-compliant manufacturing system can reassure regulators, healthcare stakeholders, and patients that medicines are safe, reliable, and consistent.
Inotek: Your Strategic Partner in GMP-Compliant Pharma Facility Rebuilding in Syria
The complexities of rebuilding Syria’s pharmaceutical manufacturing capacity demand specialized expertise and compliance-focused execution. This is where Inotek steps in as your strategic partner. We don’t just restore pharmaceutical facilities; we engineer GMP-compliant infrastructure that meets international regulatory standards, including FDA, EMA, and CDSCO, ensuring quality, efficiency, and sustainability at every stage of production.
Our comprehensive approach includes:
- Turnkey GMP Facility Construction: From conceptual design to final handover, Inotek ensures facilities are built with regulatory-grade infrastructure and meet GMP standards at every phase, ensuring quality and safety from the start.
- GMP Compliance at Every Stage: Our multi-disciplinary team covers quality compliance, engineering, project management, and equipment integration, offering a holistic “all-under-one-roof” approach that minimizes coordination challenges and compliance risks.
- Scalability and Adaptability: Whether you’re building a new facility or retrofitting an existing plant, Inotek customizes designs to meet international standards and evolving regulatory regimes, accommodating various dosage forms and capacity needs for diverse rebuilding scenarios.
By partnering with Inotek, pharma manufacturers have achieved:
- Faster facility commissioning timelines with reduced coordination hassles
- Reduction in regulatory gaps and CAPAs
- Enhanced sustainability and seamless GMP alignment
Future-Readiness: Beyond Basic Compliance
While compliance forms the foundation, successful pharmaceutical manufacturing in post-conflict settings like Syria must also address growing industry expectations around:
- Environmental sustainability in pharma operations
- Supply chain resilience for drug production and distribution
- Data integrity and digital compliance in line with evolving global regulations
At Inotek, we ensure your facility or system isn’t just audit-ready, but engineered for long-term operational excellence, capable of adapting to future challenges in the pharmaceutical sector.
Recognized among the Top 10 Pharma Turnkey Contractors & Project Consultants, Inotek helps pharma leaders design, build, and upgrade facilities that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a greenfield facility or optimizing an existing setup, Inotek ensures your project is compliant, resilient, and future-proof.
FAQs
How can GMP compliance support the rebuilding of Syria’s pharmaceutical industry?
GMP compliance is crucial for restoring Syria’s pharmaceutical manufacturing sector post-conflict. By ensuring that local production meets international standards, GMP helps Syrian manufacturers improve product quality, safety, and efficiency, reducing reliance on imports and contributing to the country’s economic recovery.
What are the key GMP requirements for pharmaceutical manufacturers in Syria?
For pharmaceutical manufacturers in Syria, GMP requirements include a robust Quality Management System (QMS), qualified personnel, controlled environments (cleanrooms, HVAC), proper sanitation, and traceable documentation. These standards ensure product consistency and regulatory compliance, essential for rebuilding trust in Syria’s local drug supply.
How does GMP compliance impact pharmaceutical manufacturing in Syria?
GMP compliance impacts Syria’s pharmaceutical industry by ensuring high-quality production. With GMP standards, Syrian pharma companies can regain regulatory approval, restore public trust, and improve access to essential medicines. This is critical for the country's long-term recovery and reducing dependence on imported drugs.
Why is infrastructure design critical for GMP-compliant pharmaceutical facilities in Syria?
Infrastructure design in Syria’s GMP-compliant pharmaceutical facilities is critical for minimizing contamination, ensuring efficiency, and supporting global compliance. Proper zoning, cleanroom setups, and validated systems in new or retrofitted plants are essential to ensuring that Syria’s local production meets safety and quality standards.
How can Inotek help pharmaceutical companies in Syria meet GMP standards?
Inotek supports pharmaceutical companies in Syria by providing turnkey solutions for GMP-compliant facilities. From design to construction and validation, Inotek ensures that all infrastructure aligns with global regulatory standards, helping Syrian manufacturers rebuild their capacity and scale up production in line with GMP requirements.



