How to Design Energy-Efficient HVAC Systems for ISO 14644 Cleanrooms in Pharma Manufacturing
December 03, 2025

Energy-Efficient HVAC Design for ISO 14644 Cleanrooms
Are your cleanroom HVAC systems draining more energy than necessary even when ISO 14644 standards are met?
In pharmaceutical facilities, HVAC systems contribute to 50–75% of cleanroom energy consumption. Even minor inefficiencies can inflate operational costs, trigger environmental monitoring (EM) deviations, and compromise compliance. As energy prices rise and regulatory expectations intensify, pharma engineering teams face the challenge of maintaining ISO-classified stability without overloading airflow, filtration, or cooling systems.
This article explains how ISO 14644 guides HVAC design and offers proven, GMP-compliant strategies to help your cleanrooms reduce energy use, improve system stability, and stay audit-ready without compromising contamination control.
Understanding ISO 14644 HVAC Requirements for Energy-Efficient Cleanrooms
ISO 14644 is the global foundation for cleanroom HVAC system design and control. For pharma teams, these are not just regulatory checkpoints; they define how much air needs to be moved, how stable the environment must remain, and how much energy the system will ultimately consume.
What Each ISO 14644 Part Means for HVAC:
- ISO 14644-1: Defines airborne particle concentration limits for different cleanroom grades. These limits directly drive airflow volume requirements.
- ISO 14644-2: Sets expectations for continuous monitoring of airflow, pressure, temperature, and other parameters to ensure HVAC stability during production.
- ISO 14644-3: Details the testing methods to validate HVAC performance, including airflow visualization, filter integrity testing, and recovery time assessments.
- ISO 14644-4: Guides how cleanroom layouts and HVAC systems should be designed to ensure predictable airflow patterns, pressure cascades, and contamination control.
Complementary Global Guidelines
EU GMP Annex 1 and WHO TRS 1019 echo these principles, reinforcing the need for:
- Smooth and directional airflow
- Consistent pressure differentials
- Repeatable environmental performance under operational conditions
Airflow Rates, ACH & Pressure Cascades
Air change rates (ACH) drive most of the energy consumption in pharma cleanrooms. Higher ACH means more fan power, larger cooling loads, and higher operational costs. ASHRAE and ISPE guidance encourage teams to challenge legacy ACH values and align them with actual risk rather than tradition.
Pressure cascades are equally important. Cleanrooms rely on a controlled pressure ladder, for example, Grade B higher than Grade C, to prevent contamination migration. Poorly managed pressure cascades lead to leakage, unstable airflow, and wasted energy. Annex 1 highlights that maintaining defined pressure differentials is essential for both sterility assurance and HVAC stability.
ISO Cleanroom Airflow Patterns
For critical zones, unidirectional airflow provides predictable removal of contaminants and supports Grade A conditions. In background areas, non-unidirectional airflow is used to dilute airborne particles efficiently. ASHRAE TC 9.11 and industry guidelines indicate that selecting the appropriate airflow pattern stabilizes conditions and reduces unnecessary fan loading.
Filtration Levels & Fan Energy
HEPA and ULPA filters protect product quality, but they also introduce resistance. Higher resistance means the fan must work harder. EN 1822 and ASHRAE 52.2 highlight how filter class, pressure drop, and housing design directly affect long-term energy use. Choosing filters with lower pressure drop while still meeting the required efficiency is one of the simplest ways to reduce HVAC operating energy.
Environmental Monitoring & HVAC Stability
Environmental monitoring depends entirely on HVAC stability. When temperature, humidity, airflow, or pressure drift from design values, EM trends become unpredictable. ISO 14644-2 and FDA EM guidance highlight that inconsistent HVAC conditions often lead to deviations, alarms, or contamination events. WHO TRS 1019 reinforces that a stable HVAC system helps teams maintain control without constantly adjusting dampers, valves, or setpoints.
Core Principles of Energy-Efficient HVAC Design in Pharma Manufacturing
Improving energy efficiency in pharmaceutical cleanrooms starts with first principles, not just equipment upgrades.
Foundational Strategies for HVAC Efficiency
- Zoning cleanroom spaces properly: This allows each area to be ventilated and conditioned based on risk level and occupancy.
- Controlling airflow paths: Prevents cross-contamination and ensures air moves predictably through high- and low-grade zones.
- Strengthening insulation: Reduces thermal load, especially in facilities with high-temperature differentials.
- Sealing the cleanroom envelope: Minimizes infiltration from adjacent non-clean areas, cutting down on excess conditioning demand.
Together, these design choices enable HVAC systems to maintain ISO-class cleanliness and environmental stability without relying on excessive airflow or cooling loads.
Global Standards Supporting These Principles
- ASHRAE 90.1
Promotes energy-efficient building design through strategies that reduce unnecessary HVAC consumption. - ASHRAE Laboratory Design Guide
Emphasizes the importance of precise pressurization and directional airflow control in controlled environments. - ISPE HVAC Good Practice Guide
Reinforces that clean, consistent airflow is the foundation for both contamination control and energy efficiency. - U.S. Department of Energy (DOE)
Shows how strategic HVAC layout planning and airflow optimization can reduce lifecycle energy demand.
Cleanroom HVAC efficiency isn’t just about an advanced system; it’s about getting the design fundamentals right. When zoning, sealing, and airflow control are engineered properly, energy savings follow naturally without risking GMP compliance.
Key Energy Drivers in Pharmaceutical Cleanroom HVAC
Cleanroom HVAC systems are among the most energy-intensive components in pharmaceutical manufacturing. Much of this energy use can be traced to a few predictable drivers.
High air change rates (ACH) significantly increase fan power and cooling loads. Many legacy systems use fixed ACH values that exceed actual process requirements. Oversized air handling units (AHUs) also lead to inefficient energy use, especially when airflow exceeds demand.
Duct leakage is another major contributor. Poorly sealed ducts force fans to work harder to maintain the required airflow and pressure, leading to higher energy consumption. Internal equipment heat load, particularly in sterile filling or packaging areas, further raises cooling demand. Additionally, return-air strategies that are poorly designed or unbalanced can result in unnecessary energy loss.
ISPE energy efficiency guidance and ASHRAE cleanroom HVAC design standards consistently highlight these elements as the top areas where pharma teams can gain measurable energy savings.
Air Change Optimization
Optimising air change rates is one of the most effective strategies for reducing HVAC energy consumption in cleanrooms without compromising GMP compliance.
EU GMP Annex 1 encourages facilities to define ACH based on process-specific risk rather than defaulting to outdated, overly conservative values. Similarly, FDA guidance for sterile manufacturing supports this approach as long as airflow is adequate to maintain contamination control.
Practical optimization strategies include reducing ACH during non-production hours, implementing occupied/unoccupied modes, and using controlled recirculation where permitted by regulations. These approaches allow cleanrooms to operate efficiently while preserving the environmental stability required for ISO-classified performance.
Practical Strategies for Cleanroom Energy Reduction
Cleanroom energy-saving initiatives must never compromise GMP compliance, particularly with regard to airflow stability, contamination control, and environmental consistency. Any reduction in HVAC load must continue to support ISO 14644 requirements and regulatory expectations, such as those outlined in EU GMP Annex 1.
The most effective strategies target the core energy drivers in cleanroom HVAC systems: fan power, conditioning of outside air, pressure maintenance, and filtration efficiency. Optimising these areas can lead to significant cost reductions while preserving process integrity.
ASHRAE guidance recommends targeted improvements in airflow management, thermal control, and fan efficiency for cleanroom environments. These include refining airflow paths, right-sizing equipment, and balancing pressure zones to avoid over-conditioning. ISPE’s sustainability frameworks support risk-based energy optimization, encouraging changes that enhance system performance without compromising regulatory compliance or audit readiness.
The U.S. Department of Energy (DOE) identifies energy recovery and system tuning as key interventions for high-performance cleanrooms. Techniques such as heat recovery, demand-based airflow control, and scheduled system adjustments can all contribute to reduced energy demand across the cleanroom lifecycle.
VFDs & Demand-Controlled Ventilation
Variable Frequency Drives (VFDs) help teams reduce energy waste by adjusting fan speed to match real operational demand rather than running at fixed maximum load. DOE guidance shows that VFDs can significantly cut kWh consumption when airflow changes with occupancy or production activity.
ASHRAE guidance supports their use in systems that require precise pressure control, since modulating fan speed helps maintain stable conditions in ISO-classified rooms. Demand-controlled ventilation (DCV) extends this benefit by using sensors for particle levels, pressure, or occupancy. This approach maintains compliance while preventing over-ventilation, which is a major source of energy waste in cleanrooms.
Low-Leakage Ducting & Efficient AHUs
Duct leakage forces fans to work harder to maintain the required airflow and pressure cascades. SMACNA standards highlight how low-leakage duct construction sharply reduces air loss and stabilizes room conditions.
Efficient AHUs built with tight sealing, proper insulation, and well-designed coils reduce both thermal load and fan power. Eurovent certification guidelines show how AHU efficiency improves both performance and energy stability in cleanroom-grade systems.
Efficient HEPA/ULPA Selection
HEPA and ULPA filters are essential for product protection, but they add significant resistance to the system. Selecting filters with lower pressure drop while still meeting ISO cleanliness needs is often one of the simplest ways to reduce fan energy without affecting contamination control.
Heat Recovery Systems
Cleanrooms exhaust large amounts of conditioned air, which increases heating and cooling demands. Heat recovery systems, such as energy wheels, plate heat exchangers, or sensible-only recovery units, capture energy from exhaust air and transfer it to the incoming air. ASHRAE Standard 84 shows how these systems significantly reduce heating and cooling loads, especially in facilities with high outdoor air volumes.
Smart BMS & Real-Time Analytics
A smart Building Management System (BMS) helps teams maintain tight control over airflow, temperature, humidity, and pressure, the core parameters required for ISO and GMP compliance.
By continuously analysing performance data, teams can fine-tune setpoints, detect inefficiencies early, and ensure stable GMP-compliant operation at lower energy cost.
Implementation Roadmap for ISO 14644-Compliant Energy-Efficient HVAC Systems
A structured implementation roadmap helps pharma facilities improve HVAC efficiency while maintaining ISO 14644 and GMP requirements. This approach ensures that design choices, system upgrades, and validation activities support long-term stability, compliance, and energy performance. ISPE commissioning and qualification guidelines highlight how early planning and systematic execution strengthen lifecycle reliability. ASHRAE commissioning principles emphasize the need for performance verification at key project stages.
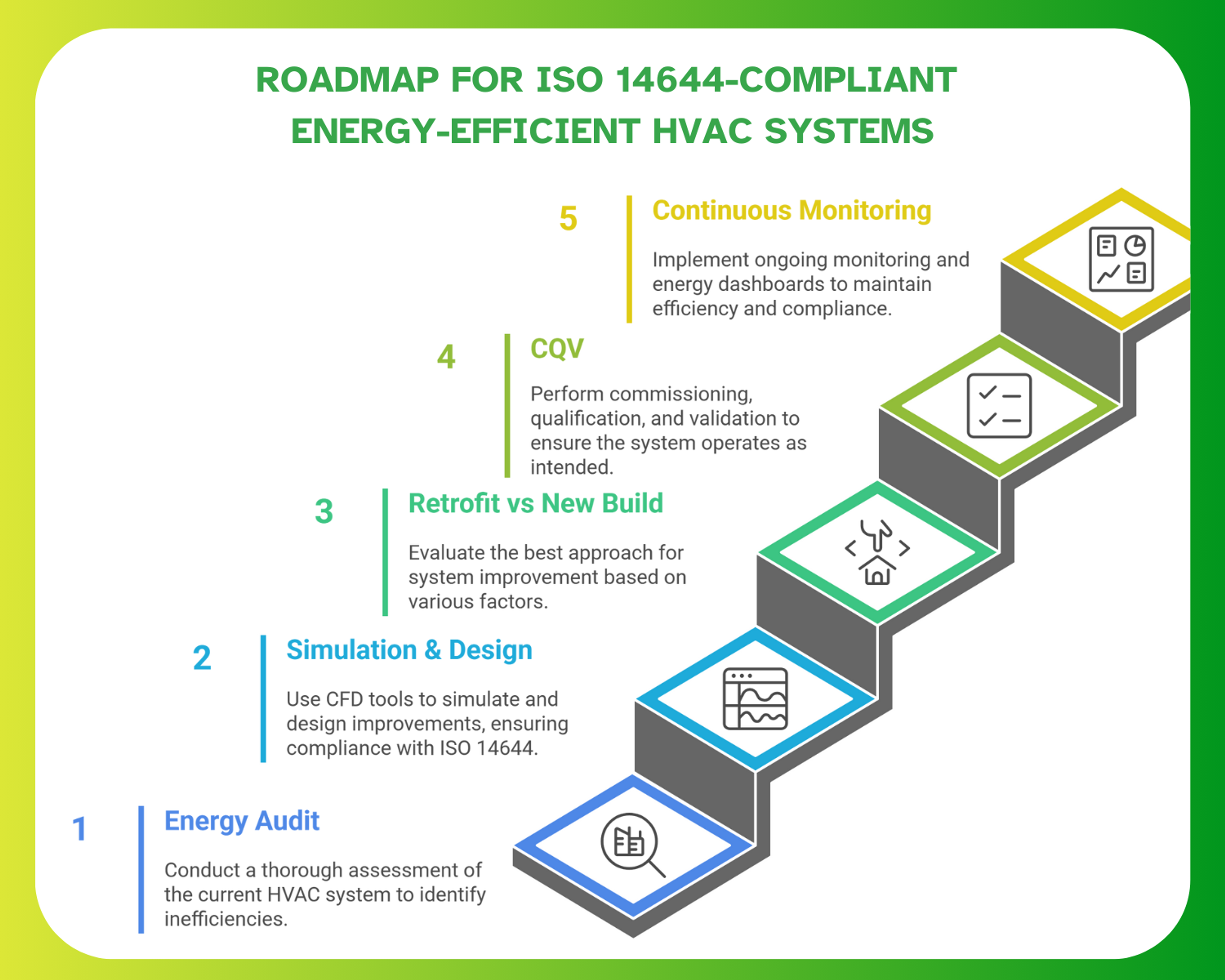
Step 1: Energy Audit & Performance Benchmarking
Energy improvement begins with understanding how the current system performs. An audit typically reviews airflow volumes, fan curves, equipment efficiency, duct leakage, and pressure stability. These baseline measurements reveal gaps such as oversized fans, excessive ACH, or air loss through poorly sealed ducts. Benchmarking also helps determine whether existing conditions meet ISO 14644 expectations or require corrective actions.
Step 2: Simulation & Design Using ISO 14644 Principles
Before implementing design changes, CFD simulations help verify airflow patterns, particle behaviour, and pressure cascades. This allows engineering teams to test proposed improvements without disrupting production. Software tools such as ANSYS provide airflow and contamination modelling tailored for controlled environments. Autodesk CFD tools also allow teams to visualize pressure zones and evaluate ISO 14644-compliant design decisions early in the process.
Step 3: Retrofit vs New Build Decision Framework
Choosing between retrofit and new build depends on system age, building condition, downtime availability, and ROI expectations. Retrofitting can be efficient when existing layouts support improved airflow, sealing, and pressure control. ISPE retrofit guidance explains how targeted upgrades, such as VFD installation, AHU replacement, or duct sealing, can deliver strong returns without full reconstruction. ASHRAE retrofit frameworks support evaluating long-term energy gains against operational impact.
Step 4: CQV (Commissioning, Qualification & Validation)
CQV ensures that the HVAC system operates as designed and meets ISO 14644 and GMP expectations. Commissioning confirms equipment performance and control accuracy. Qualifications include airflow-volume checks, pressure-cascade verification, HEPA-integrity testing, and recovery-time assessments. Validation activities guided by Annex 15 ensure that the system supports consistent cleanroom performance under operational conditions. Smoke studies confirm airflow direction and uniformity, supporting Annex 1 readiness.
Step 5: Continuous Monitoring & Energy Dashboards
Once operational, cleanroom HVAC efficiency relies on continuous monitoring. Energy dashboards help teams track long-term performance trends, detect deviations early, and optimize airflow or temperature setpoints without affecting compliance. Modern monitoring platforms support advanced analytics and automated reporting, improving audit readiness and system stability. Consistent monitoring ensures that ISO 14644 requirements remain met while preventing gradual energy drift.
How Inotek Delivers Energy-Efficient HVAC Solutions for ISO 14644 Cleanrooms
The challenges of building energy-efficient, ISO 14644-compliant cleanroom HVAC systems go far beyond equipment selection. They demand risk-based design, contamination-controlled execution, and lifecycle energy optimization, all aligned with global regulatory expectations.
This is where Inotek steps in as your strategic partner.
We don’t just integrate HVAC equipment; we engineer high-performance, GMP-aligned cleanroom environments that maintain ISO stability while reducing operational energy demands fully aligned with FDA, EMA, and CDSCO expectations.
Our comprehensive approach includes:
1. ISO 14644-Aligned Cleanroom HVAC Engineering
Designing airflow, pressure cascades, zoning, and filtration systems that maintain contamination control while improving HVAC efficiency.
2. High-Efficiency AHU & Ducting Architecture
Deploying low-leakage duct systems, optimized coil sizing, and energy-efficient AHUs that reduce fan power and thermal load across the cleanroom lifecycle.
3. Digital HVAC Control & Smart BMS Integration
Implementing advanced automation, energy dashboards, and real-time monitoring to stabilize critical parameters and reduce kWh consumption.
4. CQV-Driven Delivery & Regulatory Readiness
End-to-end Commissioning, Qualification, and Validation (CQV) support ensures the entire HVAC stack performs as designed with airflow, recovery, and pressure tests aligned with Annex 1.
5. Retrofit & Modernisation Solutions
Upgrading legacy systems with VFDs, improved filtration, duct repairs, heat-recovery add-ons, and control upgrades, delivering energy savings without structural rebuild.
By partnering with Inotek, pharma manufacturers have achieved:
• Faster and more predictable HVAC commissioning timelines
• Reduced regulatory observations linked to airflow, pressure, and EM stability
• 20–35% energy savings through targeted optimization and modernisation
• Improved equipment reliability and lower lifecycle maintenance cost
While regulatory compliance forms the foundation, modern HVAC design must address evolving industry priorities such as:
• Environmental sustainability and reduced carbon footprint
• Supply-chain resilience for critical HVAC components
• Digitally enabled monitoring and data integrity across systems
At Inotek, we ensure your cleanroom HVAC system isn’t just compliant with today’s audits; it’s engineered for long-term stability, sustainability, and operational excellence.
Recognized among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma companies design, build, and modernize cleanroom HVAC systems that meet the strictest global standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you’re planning a greenfield project or upgrading an existing facility, Inotek ensures your HVAC system is compliant, resilient, energy-efficient, and future-ready.
FAQs
What are the HVAC requirements under ISO 14644 for cleanrooms?
ISO 14644 outlines standards for cleanroom HVAC systems, including particle control (ISO 14644-1), environmental monitoring (14644-2), performance testing (14644-3), and layout design (14644-4). These guide airflow volume, pressure control, and cleanliness maintenance, which are essential for compliant pharmaceutical cleanroom operations.
Why is air change rate (ACH) optimization important in cleanrooms?
High ACH values increase HVAC energy usage. Optimising ACH based on risk, as allowed by Annex 1 and FDA guidance, helps reduce fan power and cooling load. This ensures cleanroom stability while lowering energy consumption and improving operational efficiency in pharma environments.
What is the difference between containment and comfort HVAC design?
Containment HVAC design prioritizes contamination control using pressure cascades, airflow direction, and tight sealing. Comfort cooling only controls temperature and humidity. Cleanrooms require containment-focused HVAC systems that meet ISO 14644 and GMP standards while maintaining energy efficiency and stable environmental performance.
How do VFDs and demand-controlled ventilation save energy in cleanrooms?
VFDs adjust fan speed in response to demand, reducing unnecessary energy consumption. Demand-controlled ventilation further optimizes airflow using sensors for pressure, occupancy, or particle levels. Together, they help cleanrooms maintain compliance while significantly reducing HVAC-related energy costs over time.




