Sterile Facility Setup in Iraq: Step-by-Step Guide for GMP-Compliant Pharma Projects
October 22, 2025

Sterile Facility Setup in Iraq: A Practical Roadmap for Pharma-Construction Teams
Ever wondered what it takes to build a world-class sterile facility setup in Iraq? From navigating heat and dust to meeting GMP standards, the process demands precision at every step. This article breaks down each stage from site selection to qualification, offering pharma leaders and project teams a clear, actionable roadmap for creating compliant, high-performing sterile facilities tailored to Iraq’s unique conditions.
The Growing Need for Sterile Facilities in Iraq’s Pharma Sector
Iraq is steadily positioning itself as a key hub for pharmaceutical production in the Middle East. With increased government support for healthcare reforms and incentives for local manufacturing, demand for GMP-compliant sterile facilities is growing. Pharmaceutical investors and EPCM (Engineering, Procurement, Construction, and Management) firms see this as a unique opportunity to expand capabilities while addressing the shortage of domestic sterile drug production.
This guide provides a detailed roadmap for sterile facility setup in Iraq, outlining the technical, regulatory, and logistical requirements. It covers every stage from site selection and design to commissioning, validation, and handover, giving project teams actionable insights on timelines, contamination controls, and Iraq-specific execution challenges.
Understanding the Strategic Importance of Sterile Facility Setup in Iraq
Iraq has launched a major initiative to localize drug manufacturing by building a dedicated pharmaceutical city in southern Baghdad, backed by over US$500 million in investment and partnerships with leading global drug companies.
The project includes five advanced manufacturing units, from biosimilar medicines and plasma-derived therapies to sterile production and medical textiles, aimed at cutting reliance on imports and accelerating technology transfer.
Sterile facilities are essential for the local production of injectables, ophthalmic products, and biologics segments that currently rely heavily on imports. By establishing local sterile manufacturing units, companies not only reduce dependency on regional suppliers but also gain strategic proximity to Iraq’s growing hospital network and tender-based procurement system.
For EPCM and pharma-construction companies like Inotek, Iraq represents a maturing market where technical precision, compliance knowledge, and local adaptability directly influence project success.
A Step-by-Step Journey: From Site Selection to Facility Hand-Over
Building a sterile facility setup in Iraq is a multi-phase process that demands regulatory awareness, engineering coordination, and contamination control planning.
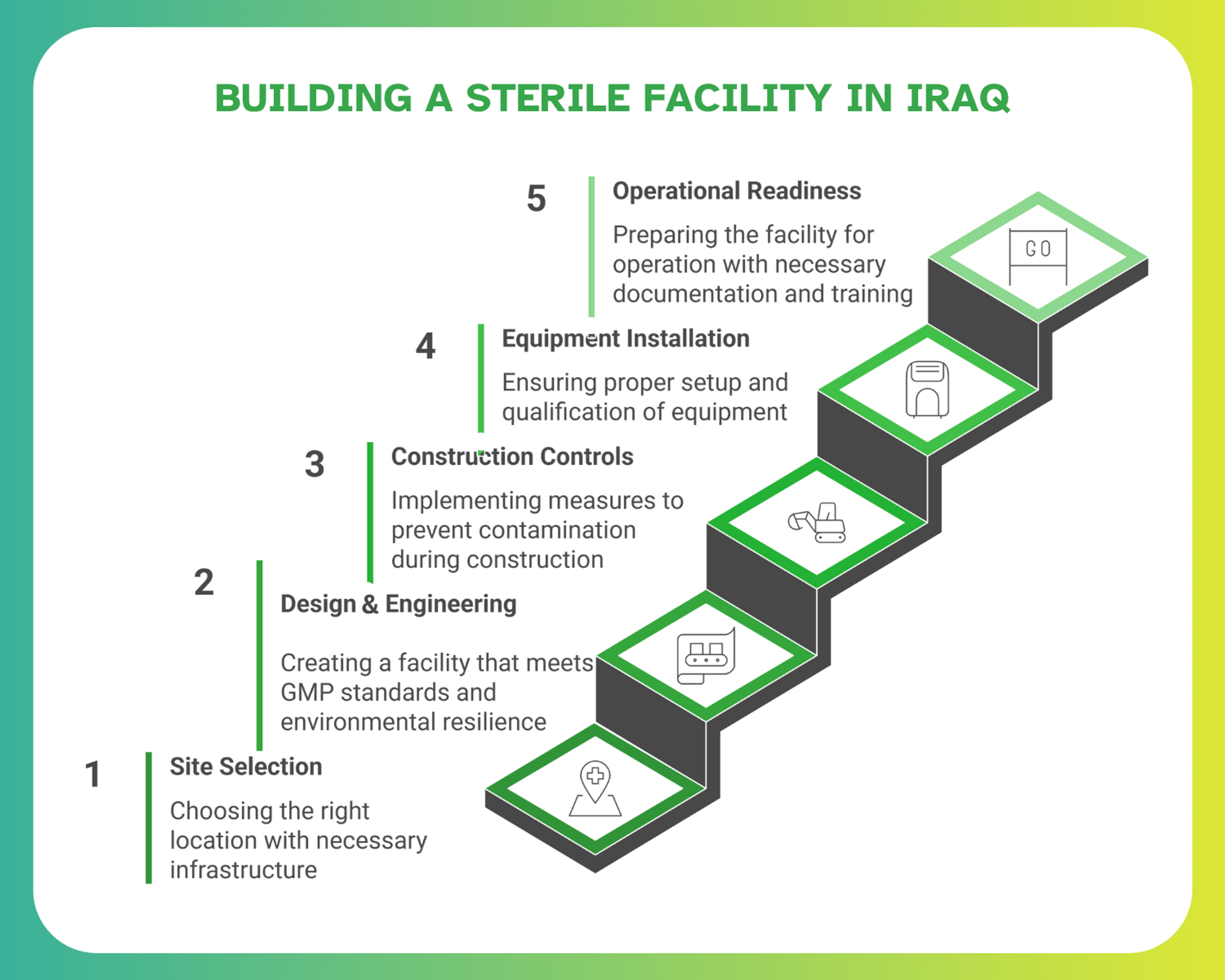
Site Selection & Feasibility
Iraq’s regional climate and infrastructure require site-specific feasibility planning. The key challenges include energy reliability, water quality, and logistics near Baghdad, Basra, and Erbil, the country’s emerging pharma zones. A feasibility study should assess not only technical viability but also regulatory zoning permissions, seismic safety, and local contractor availability.
Key Considerations for the Site in Iraq
- Location near pharma clusters (e.g., Baghdad Industrial City, Al-Zubair, and Basra Free Zone).
- Proximity to highways, airports, and seaports for import efficiency
- Consistent power and water supply, with backup energy plans.
- Local availability of HVAC, validation, and instrumentation specialists.
- Awareness of Iraq’s import and customs clearance protocols for machinery.
Design & Engineering of a Sterile Facility
Designing a sterile facility setup in Iraq requires balancing GMP compliance with environmental resilience. The HVAC system must handle 45–50°C outdoor temperatures while maintaining controlled cleanroom conditions inside. Designs should follow ISO 14644, Annex 1 of EU GMP, and WHO TRS 961 guidelines.
Cleanroom Layout & Pressure Cascade Structure
The layout should ensure strict segregation between Grade A/B clean zones and Grade C/D support areas. Pressure cascade design (e.g., +15 Pa between adjacent rooms) helps prevent cross-contamination. Material transfer airlocks and interlocks maintain directional flow integrity.
Equipment & HVAC Specification for Iraq’s Climate
A well-designed HVAC system ensures stable, cleanroom conditions despite Iraq’s hot and dusty climate. Proper equipment selection balances energy efficiency, temperature control, and contamination prevention to maintain compliance year-round.
- Dual-stage filtration (G4 + H14 HEPA) with automatic pressure drop monitoring.
- Energy-efficient AHUs with redundant chillers.
- Corrosion-resistant ducts to withstand dust and heat.
- Integration of Building Management Systems (BMS) for continuous monitoring.
Construction, Installation & Validation Planning
During construction, sterile integrity must be protected at every stage. Iraq’s desert environment introduces risks like dust infiltration, which requires stringent site controls.
Cleanroom Construction Controls: Preventing Microbial & Particulate Contamination
Maintaining sterility during cleanroom construction is critical for GMP compliance. Cleanroom controls minimize airborne particles, prevent microbial growth, and ensure structural integrity before qualification testing begins.
- Use of non-shedding materials (e.g., epoxy-coated aluminum panels).
- Controlled access zones with differential pressure monitoring.
- Real-time particulate sampling during installation.
- Compliance with ISO 14644-4 (design, construction, and start-up requirements).
Equipment Failures & Cross-Contamination Risks in Installation
Proper installation control prevents contamination during the transition from construction to operation. Addressing potential equipment failures early helps ensure sterile integrity and compliance throughout facility commissioning.
- Equipment qualification should follow risk-based validation principles.
- Cross-contamination controls include separate air-handling units (AHUs) for critical areas.
- Preventive maintenance schedules should be integrated into early commissioning phases.
Operational Readiness: Commissioning & Qualification
Operational readiness ensures that all systems, utilities, and controls work as per the validation protocol.
Documentation & Qualification Packages (DQ/IQ/OQ/PQ)
Each qualification phase must have supporting test protocols. Example:
- DQ: Review of equipment and design specs vs GMP.
- IQ: Verification of installation (utilities, safety interlocks, HEPA filter integrity).
- OQ: Testing operational parameters and alarms.
- PQ: Simulated batch production to verify environmental consistency.
Maintenance & Robustness: Long-Term Operation in Iraq
Local training programs are essential due to the limited availability of validation engineers. Establishing an on-site calibration and maintenance cell helps maintain uptime. Remote monitoring through BMS supports predictive maintenance and trend analysis.
Risk-Based Mitigation of Contamination and Equipment Challenges
Iraq’s climate and infrastructure make contamination control a continuous challenge. A robust quality risk management (QRM) framework must address both microbial and particulate threats.
Microbial Contamination: Design, Construction & Operational Controls
Controlling microbial contamination is vital to ensure product sterility. Effective design, construction, and operational protocols reduce microbial load, safeguard manufacturing processes, and uphold GMP compliance in Iraq’s challenging environmental conditions.
- Use closed systems where possible.
- Incorporate VHP (Vaporized Hydrogen Peroxide) sterilisation for decontamination.
- Schedule environmental monitoring per ISO 14698.
- Train personnel on aseptic techniques with validation logs.
Particulate Matter Contamination: Prevention Strategy
Preventing particulate contamination is crucial in sterile manufacturing. Effective surface design, air filtration, and regular monitoring help maintain cleanroom standards and protect product quality in Iraq’s demanding environmental conditions.
- Apply anti-static, smooth finishes (PVC or epoxy) on all surfaces.
- Use HEPA-filtered air showers for personnel entry.
- Continuous monitoring using laser particle counters.
Cleanroom & Equipment Failures: Risk Management Approach
Managing cleanroom and equipment failures is essential for maintaining sterility and operational efficiency. Early detection, predictive maintenance, and redundancy ensure uninterrupted compliance and consistent production in Iraq’s pharma facilities.
- Predictive maintenance based on vibration and filter pressure data.
- Dual-sensor monitoring for temperature and humidity.
- Critical utilities (WFI, HVAC, compressed air) should have redundant loops.
Cross-Contamination: Zoning, Flow & Process Separation
Managing cross-contamination is vital to protect product integrity and operator safety. Effective zoning, controlled flow, and equipment segregation help maintain sterile boundaries within Iraq’s pharmaceutical production environments.
- Use of segregated HVAC systems for antibiotic and cytotoxic zones.
- Clearly marked unidirectional personnel/material flow maps.
- Automated door interlocks to enforce flow discipline.
Project-Management Best Practices for Iraq Setup
Strong governance and local partnerships determine success in Iraq’s emerging pharma sector.
Contractual & Vendor Strategy in the Iraqi Market
Selecting the right vendor and contractual model is key to project success. Iraq’s market requires balancing international GMP expertise with local execution capabilities to ensure cost-effective, compliant project delivery.
- Combine global sterile design expertise with local EPC contractors familiar with Iraq’s safety norms.
- Include GMP-linked milestones in EPCM contracts (e.g., cleanroom readiness certification before mechanical completion).
- Ensure all materials have verifiable certification (EN/ISO/GMP documentation).
Logistics, Imports & Infrastructure Challenges in Iraq
Efficient logistics and infrastructure planning are vital for timely project delivery in Iraq. Import approvals, port delays, and energy challenges demand proactive planning to avoid costly disruptions in sterile facility setup in Iraq.
- Import timelines can extend to 6–8 weeks; engage customs brokers early.
- HVAC and validation equipment may require MoH import approvals.
- Develop alternate logistics through Basra and Umm Qasr ports to avoid delays.
- Plan for 10–15% extra lead time due to border and power disruptions.
Stakeholder Alignment & Governance Structure
Strong stakeholder alignment ensures accountability, transparency, and efficiency. Governance frameworks define clear roles, streamline decision-making, and enhance coordination between clients, contractors, and regulatory bodies throughout sterile facility setup in Iraq.
- Implement RACI-based governance (Responsible, Accountable, Consulted, Informed).
- Weekly coordination calls between the client, EPCM, and validation teams.
- Monthly progress audits aligned with GMP deliverables.
Partnering with Inotek for Sterile Facility Setup in Iraq
The complexities of delivering a sterile facility setup in Iraq demand specialized expertise, robust regulatory execution, and local infrastructure awareness. This is where Inotek steps in as your strategic partner. We don’t just build facilities; we engineer GMP-compliant, climate-resilient sterile manufacturing sites fully aligned with EU GMP Annex 1, ISO 14644 clean-room standards, and Iraq’s emerging pharma industrialisation push.
Our comprehensive approach includes:
GMP-Compliant Design & Engineering:
Translating client requirements into fully validated, audit-ready sterile facility designs. Inotek’s engineering teams integrate EU GMP Annex 1, ISO 14644, and WHO TRS 961 standards to ensure optimal cleanroom zoning, material flow, and contamination control from concept to construction.
HVAC System Design & Environmental Control:
Delivering robust HVAC systems engineered for Iraq’s hot, dusty conditions. Each system balances energy efficiency, particulate filtration (G4 + H14 HEPA), and pressure cascade integrity to sustain controlled environments and long-term sterility.
Commissioning, Qualification & Validation (CQV):
Executing complete CQV life-cycle documentation DQ, IQ, OQ, PQ, ensuring every critical utility and process system meets regulatory and performance benchmarks before handover. Our validation experts help clients achieve first-time audit success.
Regulatory & Documentation Readiness:
Supporting clients with compliance documentation aligned with Iraqi MoH, WHO, and EMA expectations. From SOP creation to qualification records, Inotek ensures your sterile facility remains fully inspection-ready at every stage.
By partnering with Inotek, pharma manufacturers in Iraq can achieve:
- Reduced commissioning schedules despite import/logistics bottlenecks.
- Fewer GMP deviations post-qualification due to risk-based design and local execution.
- Improved resilience to Iraq’s environmental and power challenges with energy-efficient HVAC and redundant utilities.
At Inotek, we ensure your facility isn’t just audit-ready, but engineered for long-term operational excellence in Iraq’s evolving pharma ecosystem.
Recognized among the Top-10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma leaders design, build, and upgrade sterile facilities in Iraq that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a green-field sterile manufacturing facility in Iraq or optimising an existing site, Inotek ensures your project is compliant, resilient, and future-proof.
FAQs
What are the key steps in a Sterile Facility Setup in Iraq?
A sterile facility setup in Iraq involves site selection, design, construction, equipment installation, commissioning, validation, and handover. Each stage integrates GMP standards, HVAC design, contamination control, and qualification testing to ensure compliance and reliable sterile manufacturing operations.
How long does it take to complete a sterile facility setup in Iraq?
A complete sterile facility setup in Iraq typically takes 18–22 months. The timeline covers feasibility, design, construction, and validation phases, with additional time for import logistics, regulatory approvals, and environmental conditioning suited to Iraq’s climatic and infrastructure conditions.
Which GMP and ISO standards apply to sterile field setup in Iraq?
The sterile field setup in Iraq follows EU GMP Annex 1, WHO TRS 961, and ISO 14644 cleanroom classifications. These standards guide HVAC design, pressure cascade structures, contamination control, and validation documentation to ensure aseptic compliance and audit readiness.
Why choose Inotek for your sterile facility construction and validation projects in Iraq?
Inotek delivers turnkey sterile facility projects in Iraq by combining global GMP expertise with local execution excellence. Their team ensures Annex 1 compliance, validated HVAC performance, and risk-based commissioning, helping pharma clients achieve faster, audit-ready, and sustainable production outcomes.




