Publisher
India Pharma Outlook
published at
October 8, 2025
What is a cGMP Certificate? Why It Matters and How to Obtain One
Is your pharmaceutical facility truly inspection-ready or just operational?
In today’s tightly regulated industry, even the most minor oversight in quality systems or facility design can lead to costly delays, rejected batches, or failed audits. That’s why every leading pharmaceutical manufacturer invests in one key assurance, cGMP certification.
A Current Good Manufacturing Practice (cGMP) certificate confirms that a facility consistently meets global standards for product quality, safety, and reliability. It isn’t just a regulatory checkbox; it’s the benchmark that determines whether a plant can legally produce and export medicines.
For pharmaceutical companies and their engineering or construction partners, this certification represents far more than compliance; it’s proof of operational excellence, regulatory confidence, and readiness for global market access.
What is a cGMP Certificate?
A cGMP certificate is proof that a pharmaceutical manufacturing site meets regulatory expectations for quality, safety, and reliability. It is not a universal document, but rather a formal acknowledgement issued by regulators such as the FDA, EMA, or CDSCO, or by accredited third-party auditors.
The certificate indicates that the facility, its equipment, documentation, and processes comply with the latest Current Good Manufacturing Practice (cGMP) standards. For manufacturers, it means eligibility for regulatory inspections and product approvals. For construction and EPCM partners, it reflects that the plant was designed and built with compliance built in.
Definition of cGMP vs GMP
GMP (Good Manufacturing Practice) establishes minimum standards for ensuring the quality of medicines. cGMP (Current Good Manufacturing Practice) emphasises that manufacturers must use the latest technologies, methods, and controls rather than relying on outdated practices.
- GMP = baseline quality compliance.
- cGMP = compliance with continuous improvement.
This difference is crucial in global markets, where regulators expect plants to remain aligned with evolving standards. Hence, the debate of GMP vs cGMP often comes down to whether a facility is operating at today’s quality expectations.
Who issues a cGMP Certificate?
A cGMP certificate may be issued by:
- Regulators: FDA (US), EMA (Europe), CDSCO (India), MHRA (UK), TGA (Australia).
- International bodies: WHO GMP certification supports prequalification and procurement in global health programmes.
- Third-party auditors: Accredited agencies that assess and confirm compliance.
No single universal certificate exists. Instead, approvals vary by region, and many exporters must hold multiple certifications to serve different markets.
Scope of Certification
A typical cGMP certificate covers:
- Facilities – cleanrooms, HVAC, utilities, material flow, segregation.
- Processes – validated production steps and controls.
- Documentation – SOPs, batch records, deviation logs, and change controls.
- Quality systems – training, CAPA, internal audits, data integrity.
For pharma-construction teams, this scope highlights why design, CQV, and utilities must align with regulatory inspection needs from the outset.
Why is a cGMP Certificate Important for Pharma Companies?
Obtaining a cGMP certificate is not optional; it is critical for regulatory compliance, operational continuity, and international market access. Failure to meet cGMP standards can lead to product recalls, FDA 483 observations, warning letters, import bans, and major financial and reputational losses (e.g., cost of destroyed batches, damage to brand trust)
The following sub-areas highlight why cGMP certification is vital:
Compliance & Regulatory Approvals
- Regulatory authorities rely on cGMP compliance assessments as the foundational criterion to grant marketing authorizations, facility licensure, and export permissions. Without such compliance, product submissions may be rejected or delayed.
- Inspections by regulators (FDA, EMA, CDSCO) often verify adherence to cGMP principles. A valid audit report or certificate helps satisfy inspection criteria and respond to regulator queries or audit findings.
- Many countries and procurement agencies demand proof of GMP compliance (or equivalent cGMP) before allowing import or marketplace entry (especially in regulated pharma markets). This requirement makes certification effectively mandatory for global competitiveness.
Business Continuity & Cost Savings
A facility that operates under cGMP standards reduces several financial and operational risks:
- Batch failures: Controlled environments, validated processes, and rigorous monitoring reduce risks of contamination, process deviations, or out-of-specification events that can invalidate entire batches. The GMP guidelines emphasise that prevention is more reliable than catching defects later.
- Downtime & rework: If non-compliance is discovered late, remediation or revalidation work may necessitate line stoppages or facility shutdowns, which can impact supply schedules and revenue.
- CAPEX overruns & retrofit costs: If compliance is considered late in the project life cycle, substantial engineering, validation, or systems rework may be required, multiplying costs.
- Efficiency and waste reduction: GMP regimes encourage tighter control and monitoring of processes, which tends to reduce waste, rejects, and reprocessing expenses. Over time, these savings offset the cost of compliance investments.
Early integration of GMP planning into design and construction helps avoid hidden costs and minimize project delays in obtaining certification.
Stakeholder Confidence & Market Access
- A cGMP certificate (or recognized compliance status) conveys credibility to regulators, investors, contract manufacturers, and supply chain partners. It reassures them that the company has reliable quality practices.
- For pharma companies working with contract manufacturers, CMOs, or partners in construction and validation, cGMP compliance acts as a common benchmark of quality, fostering smoother collaboration and trust.
- In many regulated markets, having GMP/cGMP status is a gatekeeper for procurement contracts, tenders, and strategic partnerships. Without it, firms may be excluded from key business opportunities.
- For investors and board-level oversight, a cGMP certificate helps reduce perceived regulatory and operational risk, supporting strategic growth and expansion.
How to Obtain a cGMP Certificate: Step-by-Step Roadmap
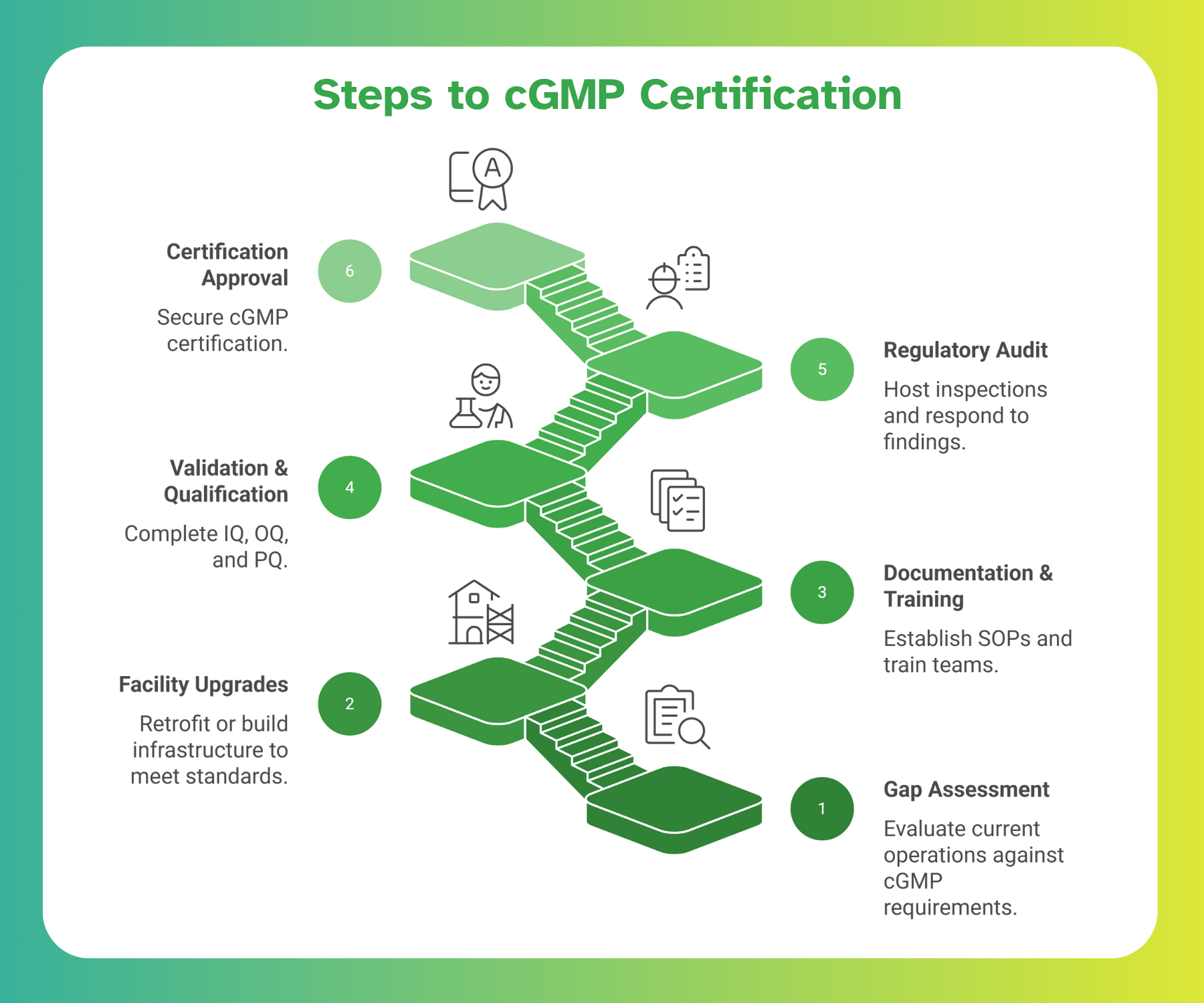
Achieving cGMP certification requires careful planning, significant investment, and meticulous execution. The roadmap typically includes:
1. Gap Assessment – Evaluate current operations against current Good Manufacturing Practice (cGMP) requirements.
2. Facility & Equipment Upgrades – Retrofit or build infrastructure to meet standards.
3. Documentation & Training – Establish SOPs and train teams.
4. Validation & Qualification – Complete IQ, OQ, and PQ.
5. Regulatory Audit & Approval – Host inspections, respond to findings, and secure certification.
In parallel, pharma teams often rely on cGMP certification online training to prepare staff for compliance requirements.
Step 1: Conducting a Gap Assessment
A structured gap analysis highlights where current systems fall short of cGMP requirements. Many companies use cGMP checklists or external consultants to benchmark readiness.
Step 2: Facility & Equipment Readiness
This step involves ensuring HVAC systems, cleanrooms, utilities, and material flow align with cGMP standards. For pharmaceutical construction project teams, this is where engineering decisions directly impact compliance outcomes.
Step 3: Documentation & Training
Certification depends on robust documentation: SOPs, validation master plans, batch records, and deviation logs. Parallel training ensures that employees understand and consistently follow procedures.
Step 4: Validation & Qualification
Regulators expect formal qualification of facilities and equipment:
- IQ – Installation Qualification
- OQ – Operational Qualification
- PQ – Performance Qualification
These stages confirm that systems function as designed under real-world conditions.
Step 5: Regulatory Inspection & Approval
Mock audits prepare the team for inspections. Agencies like the FDA may issue 483 observations, requiring corrective actions before approval can be granted. A clean inspection leads to certification or market authorization.
Common Challenges in Achieving cGMP Certification
Even with well-defined roadmaps, many organizations falter in execution. ISPE literature highlights that innovation in pharmaceutical engineering often collides with systemic barriers, and these same factors become obstacles to cGMP certification. Below are frequent non-conformities and related challenges:
Frequent Non-Conformities
- Incomplete or inconsistent documentation: Many audit findings highlight the absence of records, uncontrolled revisions, inadequate version control, or gaps in batch records and change control logs. ISPE emphasizes that strong documentation systems are fundamental to compliance.
- Insufficient staff training: Personnel who are not fully aware of cGMP principles or lack refresher training often generate errors in procedure adherence, data entry, and deviation control.
- Validation gaps in equipment or processes: Failures often occur when IQ/OQ/PQ protocols are missing, incomplete, or not aligned with risk-based quality systems.
- Weak deviation and CAPA systems: Loose tracking of deviations, delayed investigations, or weak corrective/preventive actions undermine the quality system’s credibility and may lead to recurring failures.
Best Practices for Smooth Certification
To reduce delays and increase audit success, companies should adopt these ISPE-endorsed practices:
- Start planning early, embed compliance into architectural and engineering design, and anticipate regulatory expectations during initial project phases.
- Engage EPCM and validation partners from the outset, aligning construction, utilities, and process validation oversight ensures that facility readiness is integrated (not retrofitted).
- Adopt digital QMS platforms that incorporate electronic document control, audit trails, deviation tracking, and automated workflows to enhance consistency, traceability, and regulatory inspection readiness.
- Schedule regular mock audits, internal or third-party dry-runs, to help catch gaps early. ISPE recommends frequent readiness tests to sharpen operational controls before regulators arrive.
By proactively addressing these challenges with disciplined planning, integrated teams, and modern tools, pharma firms and their construction partners can significantly streamline the path to cGMP certification.
Inotek: Your Strategic Partner in Achieving cGMP-Ready Pharma Facilities
The path to cGMP certification involves more than process validation; it begins with a solid facility foundation. The complexities of building and maintaining a cGMP-compliant pharmaceutical plant demand specialised expertise, risk-based design, and continuous alignment with global standards. This is where Inotek steps in as your strategic partner.
We don’t just construct buildings; we engineer compliance, qualification, and long-term performance into every facility, aligning it with the expectations of the FDA, EMA, WHO, and CDSCO.
Our Comprehensive cGMP Readiness Approach Includes:
Facility Design & Engineering: Turnkey solutions for cleanrooms, HVAC zoning, utilities, and process flow that meet ISO 14644 and GMP Annex 1 standards, reducing rework and downtime.
CQV (Commissioning, Qualification, Validation): Robust IQ/OQ/PQ frameworks ensure smooth audit readiness and faster regulatory approvals.
Retrofits & Expansion Projects: Modernise existing plants for cGMP compliance with minimal disruption to live operations.
Digital Documentation & Quality Systems: Integrated project documentation and validation records designed for easy inspection and lifecycle traceability.
Proven Outcomes for Pharma Clients
By partnering with Inotek, pharmaceutical manufacturers have achieved:
- Up to 30% faster facility commissioning and regulatory approval timelines.
- Fewer audit findings and reduced CAPA closures through design-integrated compliance.
- Optimised energy and utility performance, supporting both GMP and sustainability goals.
Future-Ready, Beyond Compliance
While cGMP compliance is the foundation, modern pharma facilities must also anticipate evolving expectations around:
- Sustainability and energy efficiency
- Data integrity and digital quality systems
- Operational flexibility for multi-product manufacturing
At Inotek, we ensure your facility is not only audit-ready but also engineered for long-term resilience and excellence in a competitive global market.
Recognised among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma leaders design, build, and upgrade facilities that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you’re planning a greenfield project or optimising an existing facility, Inotek ensures your investment is compliant, efficient, and future-proof.
FAQs
What does a cGMP certification mean for a pharmaceutical facility?
A cGMP certification confirms that a pharma facility follows Current Good Manufacturing Practice standards in equipment, documentation, and quality systems. It ensures regulatory compliance, consistent product quality, and readiness for inspections by the FDA, EMA, or WHO.
How is cGMP different from GMP in pharmaceutical manufacturing?
The main difference between GMP and cGMP lies in the “Current.” cGMP mandates continuous improvement using new technologies, digital monitoring, and updated regulatory practices. At the same time, GMP focuses on traditional process compliance and documentation.
What are the key steps to obtain a cGMP certificate?
Obtaining a cGMP certificate involves five stages: gap assessment, facility design upgrades, validation (IQ/OQ/PQ), training and documentation, followed by a formal regulatory audit from agencies such as the FDA, EMA, or WHO.
How long does it take to get a cGMP certification for a new plant?
The timeline for cGMP certification depends on project scope. New facilities typically require 12–24 months, covering design, validation, and audits. Using digital QMS tools and early EPCM coordination can reduce delays.



