Publisher
Inotek
published at
November 5, 2025
Vaccine Facility Setup in Saudi Arabia: Roadmap for Pharma Builders
Saudi Arabia is making significant strides toward vaccine self-sufficiency, with a USD 133 million vaccine manufacturing facility under construction in Sudair Industrial City. Spread across 42,000 m² and expected to create around 150 jobs, this facility marks a major step toward reducing vaccine import dependency and building regional biomanufacturing capacity.
For pharma-construction and EPCM firms like Inotek, this initiative highlights a new frontier in designing, building, and validating GMP-compliant vaccine plants that meet Saudi regulatory standards while aligning with Vision 2030’s localisation and innovation goals.
Project Snapshot: Scale & Purpose
This vaccine manufacturing facility represents a significant step in Saudi Arabia's push for self-sufficiency in vaccine production. Located in Sudair Industrial City, it is designed to support national health goals and international vaccine exports.
Where It’s Built: Sudair Industrial City
The new vaccine manufacturing facility in Sudair Industrial City sits about 120 km north of Riyadh. Sudair offers world-class infrastructure, access to highways, and proximity to major logistics hubs, a key factor for cold-chain and raw-material movement in vaccine production. The site falls under the Saudi Authority for Industrial Cities and Technology Zones (MODON), ensuring streamlined land allocation and utilities integration..
Investment, Scale & Jobs
With an investment of approximately USD 133 million (SAR 500 million), the project spans 42,000 m² and will employ approximately 150 people upon operationalization. The facility is being developed by the Vaccine Industrial Company (VIC), a joint venture aimed at positioning Saudi Arabia as a vaccine-production hub.
Vaccine Range & Export Plans
Production will include influenza, COVID-19, pneumococcal, meningitis, and rotavirus vaccines, supporting national immunization programs and export plans. Around 20 percent of production is targeted for international markets, underscoring Saudi Arabia’s regional export ambitions.
Why It Matters for Saudi Arabia
Saudi Arabia’s investment in local vaccine production aligns with national objectives to enhance self-sufficiency, reduce import dependency, and strengthen its biotech sector. This facility plays a key role in Vision 2030 and regional healthcare growth.
Boosting Local Vaccine Production
Saudi Arabia currently imports most of its vaccines. The new plant aims to reduce reliance on imports, improve national health security, and enhance local production capabilities, a key element of the country’s localisation policy in the pharmaceutical sector.
Tied to Vision 2030 Goals
This facility directly supports Vision 2030’s goals to build Saudi Arabia’s biotech and life sciences sector. By developing vaccine self-reliance and technology-transfer capabilities, the country is positioning itself as a biopharma innovation hub for the Middle East (GlobeNewswire).
Economic Impact & Growth Opportunities
The Saudi vaccine market is projected to grow at a 4.9 % CAGR through 2034, fuelled by innovation and localization policies. The Sudair project is expected to anchor export-led growth, with vaccines supplied to Gulf and African markets.
What It Means for Pharma Builders
The establishment of a vaccine manufacturing facility in Saudi Arabia presents unique opportunities and challenges for pharma-construction firms. From GMP compliance to project phasing, firms need to adapt their strategies to meet Saudi regulatory standards and project timelines.
Design & GMP Build Essentials
Vaccine plants require precise design standards, including GMP-compliant clean rooms, containment systems, WFI (Water for Injection), HVAC validation, and aseptic utilities integration. EPC and project-management teams must ensure modular construction, automation readiness, and microbial control across all manufacturing zones.
Regulatory & Supply-Chain Risks
Local contractors may lack GMP certification experience, requiring strong qualification oversight, vendor audits, and validation documentation (DQ/IQ/OQ/PQ). Logistics delays or material import restrictions can also impact build schedules.
Beyond regulatory complexity, vaccine facility projects in Saudi Arabia face broader challenges- including a shortage of highly skilled biopharma engineers, high operational costs, and a continued reliance on technology transfer from international partners. These hurdles often delay project timelines and affect cost efficiency if not addressed strategically.
Sudair Infrastructure Edge
Sudair’s integrated utilities, logistics corridors, and zoning simplify industrial setup. Access to Riyadh’s skilled workforce and MODON’s managed facilities enhances execution reliability.
Roadmap: From Budget to Build
The setup of a vaccine manufacturing facility in Saudi Arabia involves meticulous planning and precise budgeting. This roadmap highlights critical steps, from site acquisition to operational ramp-up, ensuring that pharma-construction firms meet timelines and budget targets.
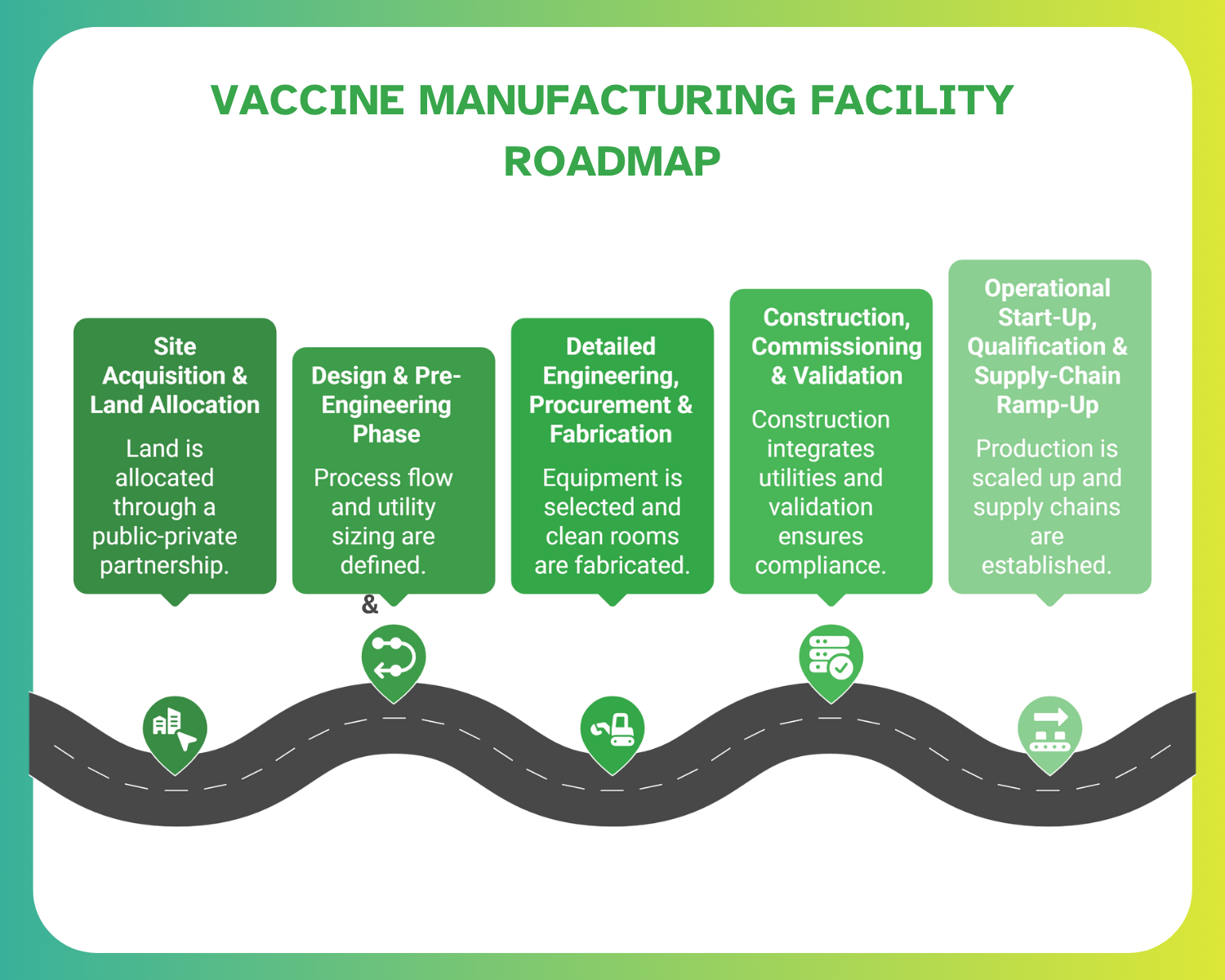
Site Acquisition & Land Allocation
Land was jointly allocated by MODON and the Vaccine Industrial Company (VIC) for the Sudair project, reflecting a strong public-private partnership. Firms must align early with MODON’s infrastructure provisions and compliance framework.
Design & Pre-Engineering Phase
Pre-engineering covers process flow definition, utility sizing (WFI, HVAC, compressed air), and clean room zoning. Early coordination with process-equipment suppliers helps ensure scalability and compliance with validation requirements.
Detailed Engineering, Procurement & Fabrication
This phase involves equipment selection, utility integration, and clean-room fabrication. Many firms opt for modular construction to shorten lead times, a model supported by KeyPlants’ agreement with KSA for modular vaccine facilities.
Construction, Commissioning & Validation
Construction integrates HVAC, WFI, autoclaves, and sterile processing areas, followed by commissioning and qualification (IQ/OQ/PQ). Detailed documentation ensures compliance with SFDA and WHO prequalification requirements.
Operational Start-Up, Qualification & Supply-Chain Ramp-Up
Final steps include operator training, batch validation, and gradual scale-up of vaccine production. Supply-chain ramp-up aligns with export certification and technology-transfer milestones.
How Inotek Elevates Vaccine Facility Setup in Saudi Arabia
Inotek plays a pivotal role in the successful setup of vaccine manufacturing facilities in Saudi Arabia by bringing together expertise in design, engineering, and project management. Their commitment to GMP compliance, regulatory excellence, and alignment with local markets helps pharma-construction firms navigate the complexities of vaccine plant setups efficiently.
Technical Design & Process Engineering Expertise
Inotek provides comprehensive expertise in designing vaccine facilities that meet the highest standards. Their capabilities include:
- Process design optimization for efficient and safe vaccine production.
- Clean-room architecture to ensure aseptic conditions throughout the facility.
- HVAC system designs that meet GMP requirements, ensuring proper air quality, temperature, and humidity control.
- Aseptic utilities integration, such as Water for Injection (WFI) and steam sterilisation, maintains vaccine integrity and safety.
Regulatory & Local Market Navigation
Navigating the regulatory environment is critical, and Inotek excels in:
- In-depth understanding of Saudi Food and Drug Authority (SFDA) guidelines for regulatory compliance.
- Managing documentation, validation protocols, and dossier submissions to ensure smooth approvals.
- Ensuring that every phase of design, construction, and operation meets local and international standards.
- Minimizing risks and delays by aligning with both Saudi and global regulatory requirements.
Modular & Accelerated Build Capability, Technology-Transfer Support
Inotek’s approach to construction and technology transfer offers significant advantages:
- Modular construction reduces on-site construction time and ensures high build quality.
- Faster project timelines through the use of prefabricated modules for a more efficient build.
- Expertise in technology transfer for scalable production, adapting the facility for different vaccine types.
- Compliance with Zone IVB standards for vaccine stability and quality.
Technical Transfer & Regulatory Dossier Support
Inotek helps overcome Saudi Arabia’s dependency on foreign technology by partnering with regional experts who provide regulatory dossier support, including e-CTD submissions, Zone IVB stability data, BE and CPP documentation, and EU GMP facility references.
This comprehensive framework accelerates SFDA approvals, ensures data integrity, and strengthens clients’ ability to meet international market entry standards.
Local-Region Presence & Alignment with Vision 2030
Inotek’s deep understanding of the Saudi market enhances project success:
- Regional presence allows for seamless collaboration with local contractors and suppliers.
- Alignment with Saudi Vision 2030, supporting localisation and biopharma infrastructure development.
- Contribution to the local workforce through training programs and technology transfer, ensuring long-term sustainability.
- Supporting Saudi Arabia’s goal to become a leading biomanufacturing hub in the region.
Through its local strategic partnerships in the Gulf region, Inotek ensures projects benefit from on-ground regulatory insights, material sourcing efficiencies, and skilled local execution teams-helping clients align seamlessly with Saudi Arabia’s Vision 2030 localisation objectives.
Next Steps for Pharma-Construction Stakeholders
Pharma-construction and EPCM firms eyeing opportunities in Saudi Arabia's vaccine manufacturing sector should take proactive steps to ensure a smooth project journey. Key actions include:
- Conduct feasibility studies to assess project viability and identify potential risks early.
- Evaluate potential partners, including local contractors, equipment suppliers, and regulatory experts.
- Establish clear budgets to ensure financial predictability throughout the project lifecycle.
- Collaborate with local regulators to ensure alignment with Saudi SFDA and other relevant standards.
- Engage experienced EPCM firms like Inotek for technical expertise in design, construction, and regulatory navigation.
- Focus on technology transfer to ensure smooth production scaling and adaptation to future vaccine requirements.
By focusing on these early-stage actions, stakeholders can mitigate risks, ensure regulatory compliance, and accelerate the path to commercial readiness.
Bridging Saudi Arabia’s Research-to-Manufacturing Gap
Saudi Arabia’s growing R&D ecosystem still faces limited downstream manufacturing integration. Inotek bridges this gap by enabling process scale-up, technology translation, and pilot-to-commercial validation.
By supporting early-stage research institutions and biotech innovators, Inotek ensures scientific breakthroughs evolve into GMP-compliant, production-ready vaccine platforms within the Kingdom.
Inotek: Your Strategic Partner in Vaccine Facility Setup in Saudi Arabia
The complexities of building a vaccine manufacturing facility in Saudi Arabia demand specialized expertise, precise design, and strict adherence to regulatory standards. This is where Inotek steps in as your strategic partner.
We don’t just build facilities; we engineer GMP-compliant, scalable vaccine production plants into every facility we deliver- aligned with global regulatory standards such as those of the FDA, EMA, and SFDA.
Our Comprehensive Approach Includes:
- GMP-Compliant Facility Design: We design cleanrooms, aseptic utilities, and HVAC systems to meet the highest safety and quality standards, ensuring seamless vaccine production from day one.
- Regulatory Navigation and Local Compliance: With years of experience in Saudi regulatory requirements, we ensure your facility aligns with SFDA guidelines, reducing approval times and regulatory risks.
- Modular Construction for Speed & Flexibility: Our modular construction approach reduces build timelines, enabling faster delivery of facilities while maintaining flexibility for future scalability and technology transfer.
- Technology Transfer & Process Integration: Inotek supports technology transfer, enabling seamless adaptation across different vaccine types and ensuring high-quality production.
By Partnering with Inotek, Pharma Manufacturers Have Achieved:
- Faster facility commissioning timelines, ensuring quicker time-to-market for vaccines.
- Reduction in regulatory gaps or CAPAs, resulting in smoother audits and approvals.
- Enhanced sustainability and GMP alignment, meeting long-term operational goals.
Future-Readiness and Broader Context
While compliance forms the foundation, successful vaccine facility setups must also address growing industry expectations around supply chain resilience, data integrity, and environmental sustainability. At Inotek, we ensure your facility or system isn’t just audit-ready, but engineered for long-term operational excellence.
Connect with Inotek for Your Vaccine Facility Setup in Saudi Arabia
Recognized among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma leaders design, build, and upgrade facilities that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a greenfield facility or optimising an existing setup, Inotek ensures your project is compliant, resilient, and future-proof.
FAQs
What is the investment size of the vaccine manufacturing facility in Sudair Industrial City, Saudi Arabia?
The vaccine manufacturing facility in Sudair Industrial City involves a USD 133 million investment, covering a 42,000 m² site. It’s part of Saudi Arabia’s Vision 2030, aiming to boost local vaccine production, create jobs, and reduce dependency on imports.
Why is Saudi Arabia building its own vaccine manufacturing facility?
Saudi Arabia is establishing its own vaccine manufacturing facility to strengthen national health security, enhance self-sufficiency, and reduce vaccine imports. The project supports Vision 2030, driving localisation, innovation, and regional biopharma competitiveness across Sudair Industrial City and beyond.
How does Inotek support the setup of a vaccine facility in Saudi Arabia?
Inotek supports the setup of a vaccine facility in Saudi Arabia through GMP-compliant design, modular construction, and regulatory navigation. Its regional expertise ensures faster project execution, compliance with SFDA regulations, and alignment with Vision 2030’s localisation and biotech growth goals.
What are the benefits of Sudair Industrial City for vaccine facility construction?
Sudair Industrial City offers strong infrastructure, logistics connectivity, and MODON-managed utilities — ideal for pharma-construction projects. These advantages make it a preferred site for vaccine manufacturing facilities seeking regulatory readiness and efficient cold-chain access across Saudi Arabia.
How long does it take to complete a vaccine manufacturing project in Saudi Arabia?
A vaccine facility setup in Saudi Arabia typically takes 2–3 years — covering site acquisition, design, construction, and validation. Partnering with experienced firms like Inotek ensures timely delivery, cost predictability, and SFDA-compliant qualification.



