Publisher
India Pharma Outlook
published at
September 10, 2025
How Eli Lilly’s $27B Pharma Facility Investment Is Shaping the Pharmaceutical Industry's Future
Is your current pharma facility strategy ready for the next decade of drug manufacturing?
Most aren’t, and that’s the challenge.
With growing pressure on supply chains, rising demand for complex therapies such as GLP-1 injectables, and increasingly stringent GMP requirements, pharmaceutical companies can no longer rely on traditional plant models. What’s needed now are digitally enabled, future-ready facilities that can scale fast and stay compliant.
That’s exactly what Eli Lilly is building.
The company’s recent $27 billion investment across four U.S. manufacturing sites marks a pivotal moment for the industry, ushering in a new era of speed, compliance, and supply chain control.
This article breaks down:
- Why Lilly is making this investment now
- What these new facilities will include, from APIs to digital twins
- What it means for QA leaders, pharma EPC firms, and construction partners
Whether you're leading quality, overseeing engineering, or managing project delivery, this article gives you a first-mover’s view into where the pharmaceutical industry's future is headed and how to prepare for it.
Why Eli Lilly Is Investing $27 Billion in New Manufacturing Sites
Eli Lilly’s decision to spend $27 billion across multiple U.S. manufacturing locations is aimed at expanding production for high-demand therapies, improving supply chain control, and reducing reliance on overseas production
The move is both strategic and reactive, responding to current therapeutic demand, reshoring trends, and government incentives that make U.S.-based manufacturing more viable. These investments reflect a future-focused shift in how leading pharmaceutical companies plan and execute pharma facility investment strategies.
How Supply Chain Disruptions Are Driving Reshoring Initiatives
The COVID-19 pandemic made one thing clear: pharma companies cannot afford fragile supply chains. Critical components and materials delayed due to border closures, geopolitical friction, and logistics breakdowns exposed vulnerabilities that led to treatment shortages and production halts
Lilly’s investment is part of a broader reshoring trend, where manufacturers are relocating their operations closer to the end market to gain better control, traceability, and faster response times. Localising manufacturing also supports stricter compliance, quality monitoring, and direct regulatory engagement, highlighting a transformative shift in the pharmaceutical industry's future.
The Explosive Growth of GLP-1 Drugs and Injectables
Another driver is the surging demand for GLP-1 receptor agonists, such as tirzepatide, used in the treatment of diabetes and obesity. The global GLP-1 drug market is projected to grow at a 17.5% CAGR, driven by rising obesity rates, better reimbursement, and broader clinical use.
To meet this demand, Lilly is expanding sterile fill-finish lines and injectable production zones. These therapies require high-compliance facilities with validated HVAC, automated monitoring, and dedicated contamination control protocols, making advanced facility planning a top priority.
Role of Federal & State Policy Incentives in Site Selection
Public policy also plays a major role in Lilly’s decision. The U.S. government is actively encouraging domestic pharma production through tools like:
- The CHIPS Act – Incentivising domestic supply chain investment
- The Inflation Reduction Act (IRA) – Offering tax credits for clean energy and domestic production
- State-level subsidies – Such as Indiana’s and North Carolina’s offers of land, utilities, and expedited permits
These incentives reduce upfront capital risks and help fast-track complex builds, making the U.S. an attractive destination for long-term pharmaceutical facility investment.
What’s Inside the New Eli Lilly Manufacturing Sites
Eli Lilly’s $27 billion investment spans four U.S.-based manufacturing facilities, each focusing on different aspects of pharmaceutical production from active pharmaceutical ingredients (APIs) to sterile injectables and biologics. Each Eli Lilly manufacturing site is not only expanding output capacity but also embedding advanced technologies and compliance protocols at the design stage.

API Plants, Biologics, and Sterile Fill-Finish Lines
The new facilities are being built to handle a broad spectrum of products:
- API plants to support upstream chemical synthesis with high containment systems for complex molecules
- Biologics facilities focused on protein-based therapies, requiring precise environmental control and upstream cell culture tech.
- Sterile fill-finish lines tailored for GLP-1 drugs and other injectables, demanding Class A/B cleanrooms and Annex 1 compliance from day one
Each site will leverage automation, data-integrated SCADA systems, and modular utilities, reducing human error and enabling real-time GMP monitoring.
Strategic Locations Backed by State Incentives
Lilly selected Indiana and North Carolina for their skilled workforce, logistics infrastructure, and policy support. The Lebanon, Indiana, site alone is expected to receive nearly $1.9 billion in investment and is projected to create 900 permanent jobs over time.
These states offered tax credits, expedited zoning, and energy incentives, making them financially attractive while also helping speed up site approvals.
Project Timelines, Workforce Needs, and Capacity Scaling
Construction has already begun at several sites, with phased commissioning planned to begin as early as 2025. Each facility will go live in stages, starting with utility infrastructure, followed by production lines and fill-finish suites
In total, Lilly expects to create over 3,000 permanent roles and more than 10,000 construction jobs across all sites. The company is also forming local vendor and contractor partnerships to source materials and manage civil and MEP works more efficiently.
Future-Proof Facility Design: AI, Automation, and GMP Compliance
Eli Lilly’s new manufacturing facilities are not only scaling output, they are also integrating digital tools, automation systems, and high-compliance infrastructure from the ground up. This design-first approach reflects a broader industry shift toward smart pharma plants that are data-driven, modular, and compliant with the latest global standards
Using Digital Twins for Real-Time Facility Planning
Lilly is adopting AI-powered digital twins, virtual replicas of its physical plants, to plan, simulate, and optimize manufacturing operations before ground is even broken. Digital twins enable real-time visibility into process performance, equipment wear, and energy use.
Global peers, such as Pfizer and Bayer, already utilize this technology to reduce commissioning time, validate workflows, and proactively resolve design bottlenecks.
By integrating digital twins early in the design process, pharma companies can streamline qualification protocols, improve energy efficiency, and enhance GMP readiness from Day 1.
Compliance With EU Annex 1 and GMP Guidelines
The revised EU GMP Annex 1 regulations have introduced stricter expectations around contamination control, especially for sterile manufacturing. Lilly’s sites are being built to meet these standards, which emphasize:
- Dedicated cleanroom zoning
- Pressure cascade integrity
- Validated air handling and particle control
The updated Annex 1 also requires a robust Contamination Control Strategy (CCS) to be embedded in facility design, shaping how rooms are connected, pressurized, and cleaned.
This demands early coordination between QA, HVAC, and automation teams to ensure compliance without delays.
Designing for Sustainability and ESG Standards
Alongside compliance and digitisation, sustainability is a core part of Lilly’s facility design. The sites are targetingLEED certification and will include:
- Energy recovery systems for HVAC and process steam
- Water recycling units
- Design alignment with ISO 14001 for environmental management systems
Sustainable design is no longer a bonus; it’s fast becoming a bid-winning factor for large-scale pharma projects.
How Lilly’s Move Signals the Future of Pharmaceutical Industry Expansion
Eli Lilly’s $27 billion expansion reflects a broader transformation across the pharmaceutical sector. Where facility development is no longer a reactive process, but a strategic enabler of product innovation, speed-to-market, and global supply assurance. The scale and scope of these new builds are setting a benchmark that others in the industry are quickly following, shaping the pharmaceutical industry's future in significant ways.
Pharma 4.0 principles, including modularity, digitalization, and compliance by design, are becoming central to facility planning across the board. Lilly’s announcement is not an outlier; it’s a signal of where the entire industry is heading.
U.S. Pharma Facility Investment Trends in 2023–25
Between 2023 and 2025, several major pharmaceutical players have committed to U.S.-based manufacturing:
- Pfizer has invested over $11 billion in its Michigan and North Carolina sites for injectables and mRNA technologies.
- Amgen has developed a $550 million advanced biologics facility in North Carolina.
- Thermo Fisher Scientific has expanded API and fill-finish capabilities across Massachusetts and North Carolina.
However, Eli Lilly’s manufacturing site push worth $27 billion is the largest by far, both in total spend and facility diversity. It is shaping pharmaceutical facility investment expectations for future builds and raising the bar for EPC project scope and scale.
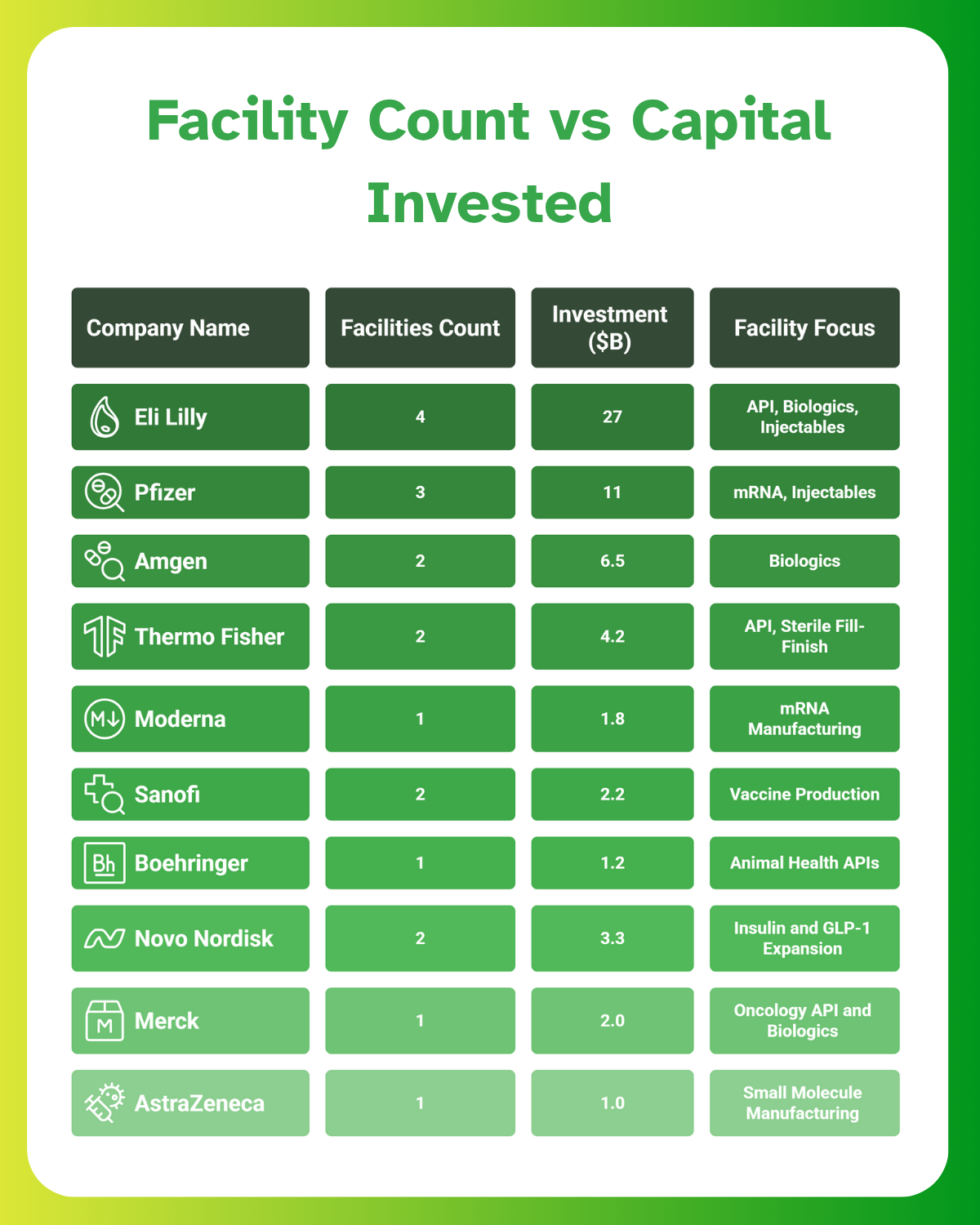
Lilly’s multi-site, multi-product strategy sets it apart, placing it at the forefront of pharmaceutical industry expansion and facility design innovation.
What It Means for Pharma Construction Firms
Eli Lilly’s multi-billion-dollar investment is not just a pharmaceutical headline; it’s a signal for engineering and project management firms that the demand for large-scale, high-speed, compliance-ready pharma infrastructure is rising fast. For PMC/EPC firms, these developments open up new opportunities and competitive requirements.
The Rise of Mega-Site Construction & Turnkey Delivery Models
Each of Lilly’s facilities is designed as a mega-site, combining API, biologics, and sterile manufacturing under one integrated campus. These types of projects require fully coordinated civil, MEP, cleanroom, and automation packages and demand contractors who can deliver all scopes under tight regulatory and construction timelines.
With aggressive go-live targets and phased rollouts, pharma companies now seek turnkey delivery partners who can align with GMP standards from day one and reduce multi-vendor complexity.
Competing for Multi-State Pharma Contracts in the U.S.
Lilly is developing multiple sites across the U.S., including Indiana and North Carolina, which require construction partners capable of navigating diverse state-level compliance codes, labour regulations, and zoning policies.
PMC firms bidding on such contracts need to:
- Harmonize compliance documentation
- Accelerate design approvals
- Manage cross-state resource mobilisation without compromising on quality or validation readiness..
How Can Firms Align With Digitally Enabled Facility Requirements
New pharma facilities are increasingly reliant on digitally integrated systems from SCADA-controlled HVAC to automated environmental monitoring dashboards. Construction teams must now deliver:
- SCADA-compatible infrastructure
- Smart HVAC zoning and control logic
- Pre-integrated panels and BMS systems that support real-time GMP deviation alerts and audit trails.
Firms unable to meet these digital expectations will struggle to compete in future-ready facility bids.
Conclusion
Eli Lilly’s $27 billion investment marks more than an expansion; it signals a shift in how the pharmaceutical industry's future is being envisioned and built. The future isn’t just about adding capacity; it’s about enabling faster drug delivery, embedding compliance from the ground up, and embracing digital systems that can scale.
For pharma leaders, QA heads, and construction partners, this is a clear signal: high-speed, tech-enabled, and regulation-ready facilities are the new standard. As more companies follow suit, aligning with this direction is no longer optional; it’s essential for staying competitive in the global drug manufacturing landscape.
How Inotek Aligns with the Pharma Facility Investment Wave
The complexities of large-scale pharmaceutical facility investment and expansion demand specialized expertise and compliance-focused execution. This is where Inotek steps in as your strategic partner.
We don’t just build infrastructure, we engineer fully compliant, digitally enabled, and audit-ready pharma environments aligned with global regulatory standards like FDA, EMA, and CDSCO.
Our comprehensive approach includes:
End-to-End Turnkey Project Delivery: From design to commissioning, we manage every scope under one GMP-governed umbrella, ensuring accountability, speed, and precision.
Cleanroom and Containment Engineering: ISO 14644-compliant cleanrooms and validated pressure cascade systems built for sterile, non-sterile, and high-potency APIs.
Integrated Automation & SCADA Systems: Real-time monitoring, alarm logic, and data integrity frameworks that streamline compliance and future inspections.
GMP Documentation & Qualification Support: Aligned with Annex 1 and WHO TRS 1019, we provide full DQ/IQ/OQ support for faster validation and audit readiness.
By partnering with Inotek, pharma manufacturers have achieved:
- 40% faster facility commissioning timelines
- Zero major audit observations across CDSCO and EU GMP inspections
- Measurable improvements in HVAC energy efficiency and zoning integrity
While compliance forms the foundation, successful pharma facility expansion and execution must also address evolving expectations around:
- Sustainability and energy recovery integration
- Resilience to supply chain and logistics disruptions
- Digital compliance and data integrity by design
At Inotek, we ensure your facility isn’t just audit-ready, but engineered for long-term operational excellence and strategic scalability.
Recognized among the Top 10 Pharma Turnkey Contractors & Project Consultants in 2022 & 2025, Inotek helps pharma leaders design, build, and upgrade facilities that meet the strictest GMP and sustainability standards.
📞 Connect with our experts today or visit www.inotek.co.in to schedule a consultation with Mr. Rohit Ochaney.
Whether you're planning a greenfield facility or optimising an existing setup, Inotek ensures your project is compliant, resilient, and future-proof.
FAQs
Why is Eli Lilly investing $27 billion in new manufacturing sites?
Eli Lilly is investing in U.S.-based facilities to meet rising demand for GLP-1 drugs, ensure GMP compliance, strengthen supply chains, and leverage government incentives.
What is the future of pharmaceutical facility design?
The future includes Pharma 4.0 concepts like digital twins, modularity, automation, sustainability, and built-in GMP and Annex 1 compliance.
How does Lilly's expansion impact pharma EPC and PMC firms?
It increases demand for turnkey construction models, integrated SCADA systems, and cross-functional project delivery capabilities with regulatory alignment.
What technologies are shaping pharma facility investment strategies?
Digital twins, cleanroom automation, SCADA integration, HVAC zoning logic, and energy-efficient utility designs are becoming industry standards.



